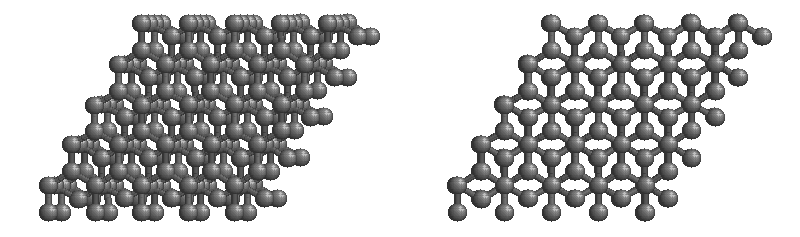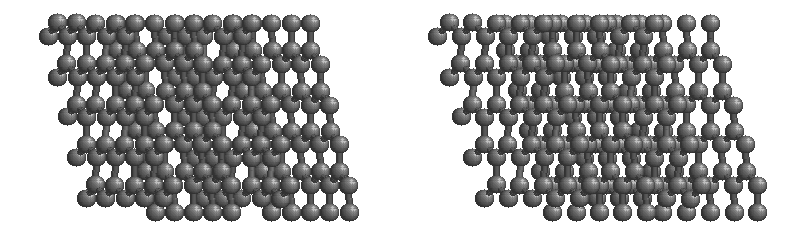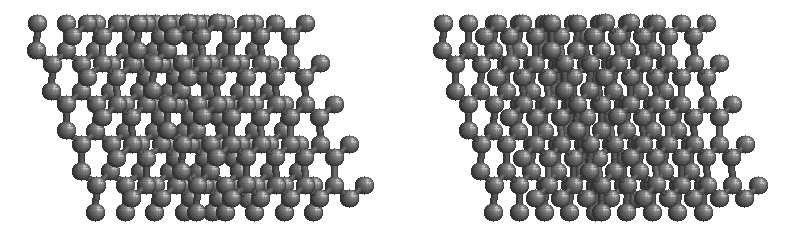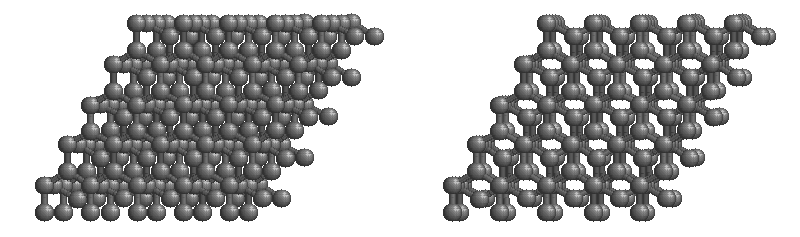Carbon network lattices
•A large number of atoms are covalently bonded together to form a 3 dimensional network lattice.
Properties of covalent network lattices:
1. exist as solids and have a very high melting point (strong covalent bonds between atoms)
2. do not conduct electricity (no charged particles)
3. are brittle, chemically inert and insoluble in water.
•Eg: diamond C, silicon dioxide (SiO2), silicon carbide (SiC) and tungsten carbide (WC).
Allotropes of carbon
Allotropes are different forms of the same element.
Carbon has 3 allotropes:
•Diamond
•Graphite
•Amorphous carbon
Diamond
•Diamond is the hardest naturally occurring substance known.
•Diamond is made up of carbon atoms that bond with four adjacent carbon atoms forming a covalent lattice.
•Properties:
-very hard
-sublimes
-non-conductive
-brittle
Diamond animation courtesy of wikimedia commons
Covalent layer lattices
•A large number of atoms are covalently bonded together to form a 2 dimensional layer lattice. Example: Graphite, C
•The layers are held together by weak dispersion forces.
•Each carbon atom is covalently bonded to three other carbon atoms.
•The network structure contains delocalized electrons.
Properties of Graphite:
1. exist as a solid with a high melting point (strong covalent bonds between atoms)
2. conducts electricity due to presence of delocalized electrons.
3.slippery to touch (weak dispersion forces between layers allow layers to slide past each other).
4.soft
Amorphous forms of carbon
•These forms have no consistent structure.
•Examples- Charcoal and carbon black.
•Can be formed from combustion
•Properties
-Conductive
-Non-crystalline
-cheap
Animation of graphite courtesy of Wikimedia commons.
•A large number of atoms are covalently bonded together to form a 3 dimensional network lattice.
Properties of covalent network lattices:
1. exist as solids and have a very high melting point (strong covalent bonds between atoms)
2. do not conduct electricity (no charged particles)
3. are brittle, chemically inert and insoluble in water.
•Eg: diamond C, silicon dioxide (SiO2), silicon carbide (SiC) and tungsten carbide (WC).
Allotropes of carbon
Allotropes are different forms of the same element.
Carbon has 3 allotropes:
•Diamond
•Graphite
•Amorphous carbon
Diamond
•Diamond is the hardest naturally occurring substance known.
•Diamond is made up of carbon atoms that bond with four adjacent carbon atoms forming a covalent lattice.
•Properties:
-very hard
-sublimes
-non-conductive
-brittle
Diamond animation courtesy of wikimedia commons
Covalent layer lattices
•A large number of atoms are covalently bonded together to form a 2 dimensional layer lattice. Example: Graphite, C
•The layers are held together by weak dispersion forces.
•Each carbon atom is covalently bonded to three other carbon atoms.
•The network structure contains delocalized electrons.
Properties of Graphite:
1. exist as a solid with a high melting point (strong covalent bonds between atoms)
2. conducts electricity due to presence of delocalized electrons.
3.slippery to touch (weak dispersion forces between layers allow layers to slide past each other).
4.soft
Amorphous forms of carbon
•These forms have no consistent structure.
•Examples- Charcoal and carbon black.
•Can be formed from combustion
•Properties
-Conductive
-Non-crystalline
-cheap
Animation of graphite courtesy of Wikimedia commons.

Carbon nanomaterials
Carbon nanomaterials are carbon allotropes with a cylindrical nanostructure.
Types:
1. Fullerenes (Buckyballs)
•The Fullerenes are an allotrope of carbon, the atoms are arranged in connecting pentagons or hexagons.
•They have a high surface area to volume ratio- better properties.
•They have a high tensile strength and high electrical and heat conductivity.
2. Graphene
• Graphene -a single-layer of graphite.
•It is soft due to weak dispersion forces between its layers.
• Good electrical conductivity
* Is an extremely strong and tough material
3. Nanotubes
•Closely related to graphene.
•They have a long hollow structure with walls formed from graphene.
•They have:
- Good electrical and thermal conductivity
-strong forces of attraction between each other
-strength
Carbon nanomaterials are carbon allotropes with a cylindrical nanostructure.
Types:
1. Fullerenes (Buckyballs)
•The Fullerenes are an allotrope of carbon, the atoms are arranged in connecting pentagons or hexagons.
•They have a high surface area to volume ratio- better properties.
•They have a high tensile strength and high electrical and heat conductivity.
2. Graphene
• Graphene -a single-layer of graphite.
•It is soft due to weak dispersion forces between its layers.
• Good electrical conductivity
* Is an extremely strong and tough material
3. Nanotubes
•Closely related to graphene.
•They have a long hollow structure with walls formed from graphene.
•They have:
- Good electrical and thermal conductivity
-strong forces of attraction between each other
-strength