|
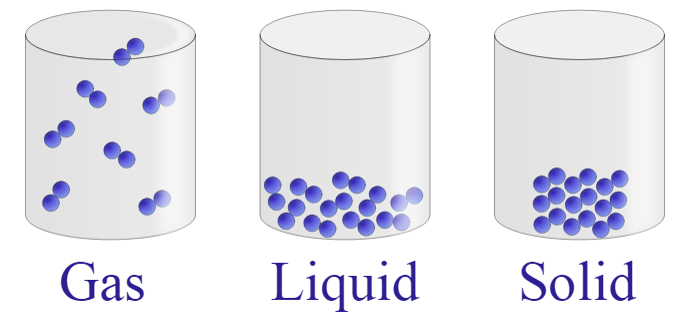
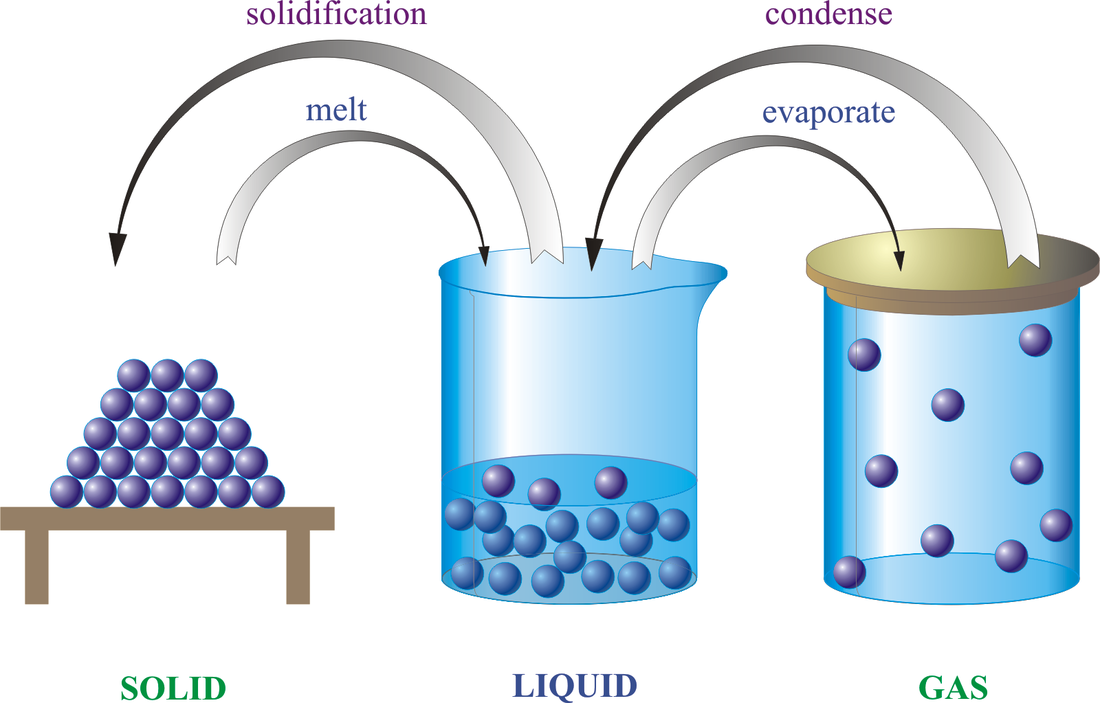
Kinetic theory of matter Key Points: 1.Matter is made up of tiny, invisible moving particles. 2.Particles of different substances have different sizes. 3.Lighter particles move faster than heavier ones at a particular temperature. 4.As the temperature rises, the particles move faster. As the temperature decreases, the particles move slower. Matter The 3 states of matter are solid, liquid and gas. SOLIDS- have restricted movement- rotation and vibration only. The particles are closely packed and held together by forces of attraction. Solids have a definite volume and shape. LIQUIDS-have less restriction of movement- they have greater rotational and vibrational movement and some translational movement. They can slide past each other. There is still enough attraction between particles to maintain a definite volume. A liquid takes the shape of the container in which it is placed. GASES- have greater freedom of movement. Gas molecules are in a constant state of random motion. They are separated by large distances and travel with great speed- they will often collide with each other. A gas will alter both its shape and volume to fill a container completely. Change of state An increase or a decrease in kinetic energy leads to a change of state. –Evaporation – conversion of liquid into gas. –Condensation – conversion of gas into liquid. –Melting – conversion of solid into liquid. –Freezing – conversion of liquid into solid. –Sublimation – direct conversion of a solid to a gas or a gas to a solid. CHEMICAL REACTIONS A chemical reaction has taken place if at least one new substance has been formed. A chemical equation can be written for chemical reactions. Eg. REACTANTS --> PRODUCTS Signs of chemical reactions include: Formation of a precipitate, formation of a gas, colour change, energy absorbed or lost. TYPES OF CHEMICAL REACTIONS *Precipitate- when an insoluble solid forms when two or more clear (not always colourless) solutions are mixed. *Exothermic- these reactions release heat- the temperature increases. *Endothermic- these reactions absorb heat- the temperature decreases. *Redox- where electrons are lost or gained in a reaction. *Combination- where two or more substances combine to form a new substance. •Eg. Carbon + oxygen --> carbon dioxide C + O2 --> CO2 *Decompostition-where a single substance is broken down into simpler substances or elements. Eg. Copper carbonate --> copper oxide + carbon dioxide CuCO3 --> CuO + CO2 *Single replacement involves removing one element or group of elements and replacing them with another element or group of elements. Eg. Copper + silver nitrate --> silver + copper nitrate Cu + 2 AgNO3 --> 2 Ag + Cu(NO3)2 *Double replacement reaction- involves two metal ions swapping to form two new compounds. (Sometimes a precipitate is formed here). Eg. Barium nitrate + Copper sulfate --> copper nitrate + Barium sulfate Ba(NO3)2 + CuSO4 --> Cu(NO3)2 + BaSO4 *Neutralisation- where an acid reacts with a base to produce a salt and water. ACID + BASE --> SALT + WATER Eg. hydrochloric acid + sodium hydroxide --> sodium chloride + water. *Acid and a carbonate base- If the base added during neutralisation contains a carbonate, then the following reaction occurs: ACID + CARBONATE BASE-->SALT + WATER+ CARBON DIOXIDE Eg. hydrochloric acid + sodium bicarbonate --> sodium chloride + water + carbon dioxide *Metal and an Acid-An acid will react with a metal to produce a salt and hydrogen gas. ACID + METAL -->SALT + HYDROGEN Eg. Hydrochloric acid + Magnesium --> magnesium chloride + hydrogen. BALANCING CHEMICAL EQUATIONS •Chemical formulas- are a shorthand representation of compounds showing the ratios of each element in the compound. eg. CuCO3 •Chemical equations- show what is reacted and what is produced along with the formula of each substance. •Eg. CuCO3 --> CuO + CO2 •Balanced chemical equations- here the numbers of atoms on one side of an equation must equal the number of atoms on the other side of the equation (conservation of mass). Zn + HCl --> ZnCl2 + H2 1 Zn 1H 1 Cl 1 Zn 2Cl 2 H Thus, to balance we need to multiply HCl by 2 to get 2 hydrogen atoms and 2 chlorine atoms. Zn + 2 HCl --> ZnCl2 + H2 Balancing chemical equations is a skill related to an area of Chemistry known as STOICHIOMETRY- which deals with the quantitative (measuring) aspects of chemistry. Physical states- symbols are added to show the physical states of the reactants and products. Solid- (s) Liquid – (l) Gas – (g) Aqueous (water) solution- (aq) |
Diagram courtesy of By Yelod - Wikimedia Commons * Yelod - Wikipedia (En) * ילוד - ויקיפדיה העברית - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=7719358
Changes of state diagram courtesy of E. Generalic, http://glossary.periodni.com/glossary.php?en=states+of+matter
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning