RATE AND EQUILIBRIUM REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-unit-3-and-4-rate-and-equilibrium-revision-11987322
COLLISION THEORY
•Collision theory; the theory used to predict the rates of chemical reactions, particularly for gases. The collision theory is based on the assumption that for a reaction to occur it is necessary for the reacting species (atoms or molecules) to come together or collide with one another. (However a collision does not guarantee a reaction).
•Not all collisions, however, bring about chemical change.
A collision will be effective in producing chemical change only if the species brought together possess a certain minimum value of internal energy, equal to the activation energy of the reaction.
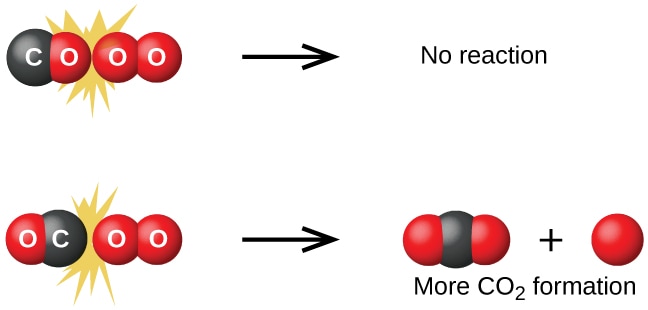
•Furthermore, the colliding species must be oriented in a manner favourable to the necessary rearrangement of atoms and electrons. Thus, according to the collision theory, the rate at which a chemical reaction proceeds is equal to the frequency of effective collisions.
•Collision theory; the theory used to predict the rates of chemical reactions, particularly for gases. The collision theory is based on the assumption that for a reaction to occur it is necessary for the reacting species (atoms or molecules) to come together or collide with one another. (However a collision does not guarantee a reaction).
•Not all collisions, however, bring about chemical change.
A collision will be effective in producing chemical change only if the species brought together possess a certain minimum value of internal energy, equal to the activation energy of the reaction.
•Furthermore, the colliding species must be oriented in a manner favourable to the necessary rearrangement of atoms and electrons. Thus, according to the collision theory, the rate at which a chemical reaction proceeds is equal to the frequency of effective collisions.
|
Image show unfavourable and favourable orientations of carbon monoxide and oxygen molecules to activate a reaction.
Image courtesy of :
|