GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
Electroplating
|
Chemicals obtained by electrolysis are often difficult to obtain by other means. Important uses of electrolysis are:
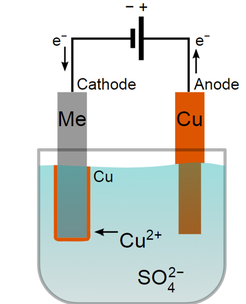
•Plating of metals on surfaces of other metals to improve appearance or prevent corrosion •Extracting reactive metals, such as Na or Al, from their ores •Industrial production of Cl, Cu, NaOH, H •Recharging car batteries and other cells. Electroplating is the deposition of a layer of metal on the surface of another metal by electrolysis. •Tin cans are an example of something that has been electroplated. Tin cans are composed mainly of steel with a thin layer of tin coating the surface. Tin corrodes slowly and prevents contact between the Iron in the can from rusting. •The object to be plated is connected by a wire to the negative terminal of a power supply; this object is regarded as the negative electrode. It is immersed in a solution which contains ions of the metal that is to form the plating. This solution is referred to as the electrolyte. In tin plating, an electrode of tin metal is connected to the positive terminal of the power supply. •When the cell is in operation the power supply acts as an ‘electron pump’ pushing electrons onto the negative electrode and removing electrons from the positive electrode. •A Cathode is defined as an electrode at which reduction occurs. In electrolytic cells, the cathode is NEGATIVE. (Because if you electrocute your cat that’s a BAD thing!!) •An Anode is defined as the electrode at which oxidation occurs. In electrolytic cells, the anode is positive. •There is an internal electric current through the cell electrolyte involving movement of cations towards the cathode and anions toward the anode. Faraday's second law of electrolysis•In order to deposit one mole of silver from a solution of Ag+ ions, just one more of electron is required:
Ag+(aq) + e- → Ag(s) •The charge on one mole of electrons is 96 500C •The charge on one mole of electrons is known as a faraday, and given the symbol F. 1 Faraday = 96 500 coulomb mol-1. •The charge on a given number of moles of electrons, n(e-), may be calculated by: Q = n(e-) x F •In order to produce one mole of a metal, one, two, three, or another whole number of moles of electrons must be consumed. •This is Faraday’s second law of electrolysis |
Copper electroplating diagram courtesy of By Torsten Henning (Multilingual Perhelion) [Public domain], from Wikimedia Commons
Faraday's first law of electrolysis•Electric charge, is given the symbol Q and is measured in coulomb. •The relationship is: charge(coulombs) = current(amps) x time(sec) Q = I x t •When electroplating an object we need to know: –How can I determine how much metal is being plated? –How long should I leave the object being plated in the electroplating cell? –What size current should I use. •We can determine the electric charge needed to be used by using this law. •We can link this to mass as the mass of metal produced at the cathode is directly proportional to the quantity of electricity passed through the cell. •This relationship is known as Faraday’s first law of electrolysis. •It may be written as: m α Q |