Fats and oils
|
Fat is a name used to describe a large number of organic compounds belonging to an even larger class of biological molecules called lipids.
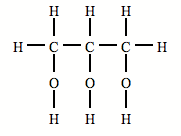
Eg. Fats, oils, waxes, steroids, cholesterol are all lipids. Most lipids are essentially non-polar and as a result are insoluble in water. Non-polar vitamins A,D,E and K are able to be dissolved in the non-polar lipids. Fats are solids. Oils are liquids. Most fats and oils are formed by a condensation reaction (esterification reaction) between a single molecule of glycerol and three molecules of fatty acids, three molecules of water is also made in this reaction. Fats and oils formed in this process are called triglycerides. Triglycerides are large, non-polar molecules and therefore insoluble in aqueous solutions. Glycerol diagram By Nilsgeek94 (Own work) [Public domain], via Wikimedia Commons
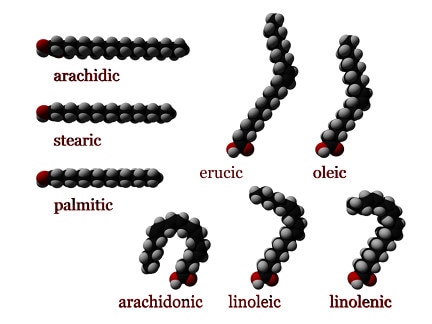
Types of fatsFats are distinguished based on the fatty acids from which they are made. •Saturated fats- made from fatty acids that contain only single carbon-carbon bonds. Saturated fats are generally unreactive and occur as waxy solids at room temperature. •Mono-unsaturated fats- made form fatty acids that contain one carbon-carbon double bond. •Polyunsaturated fats- are made from fatty acids that contain more than one carbon-carbon double bond. Polyunsaturated fats have lower melting points than saturated fats, and often occur as liquids (oils) at room temperature. They are more reactive than saturated fats. The different physical states of saturated and polyunsaturated fats are thought to arise because molecules of saturated fats can pack more closely together, resulting in stronger dispersion forces between molecules. The double bonds in polyunsaturated fats do not allow close packing. Consequently saturated fats have higher melting points than unsaturated fats. In general, animals fats contain higher proportions of saturated fats, while vegetable oils are richer in polyunsaturated fats. The fatty acids can be -cis or -trans. A cis configuration means that the two hydrogen atoms adjacent to the double bond stick out on the same side of the chain. A trans configuration means that the adjacent two hydrogen atoms lie on opposite sides of the chain. Structures of some fatty acid molecules By The original uploader was (Automated conversion) at English Wikipedia (Transferred from en.wikipedia to Commons.) [GFDL (http://www.gnu.org/copyleft/fdl.html) or CC-BY-SA-3.0 (http://creativecommons.org/licenses/by-sa/3.0/)], via Wikimedia Commons
What happens to fats in the body?Fat is mainly acted upon when it reaches the small intestine. There, it is mixed with bile from the gall bladder. Bile contains surfactants which convert fats into an emulsion of smaller fat globules. Enzymes from the pancreas catalyse the hydrolysis of fat by reversing the condensation reaction and making glycerol and fatty acids. Once these have been absorbed by the body, they are reassembled into triglycerides via condensation reactions.
|
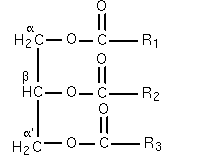
Triglyceride diagram By The original uploader was NicolasGrandjean at French Wikipedia [GFDL (http://www.gnu.org/copyleft/fdl.html) or CC-BY-SA-3.0 (http://creativecommons.org/licenses/by-sa/3.0/)], via Wikimedia Commons
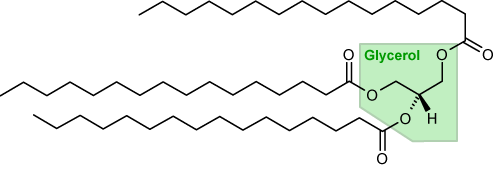
Tripalmitoylglycerol made from the condensation reaction between glycerol and 3 palmitic acid molecules, 3 water molecules are released in this reaction.
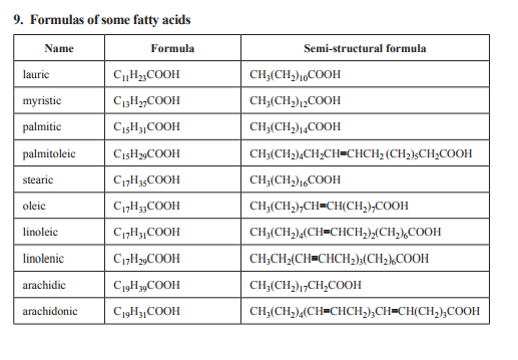
Tripalmitoylglycerol diagram courtesy of wikimedia commons. Names and formulas of fatty acids- VCE chemistry data booklet.
Cis and trans formation for Oleic acid By Edgar181 (Own work) [Public domain], via Wikimedia Commons
|