|
STUDY CARD AVAILABLE- SEE CONTACT.
FUELS AND ENERGY PLACE MAT REVISION- https://www.tes.com/teaching-resource/vce-chemistry-unit-3-and-4-fuels-and-energy-revision-11987315
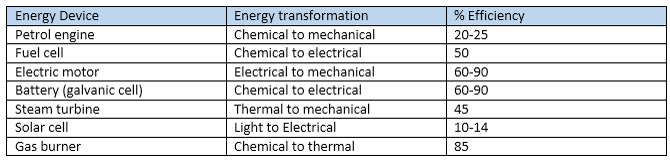
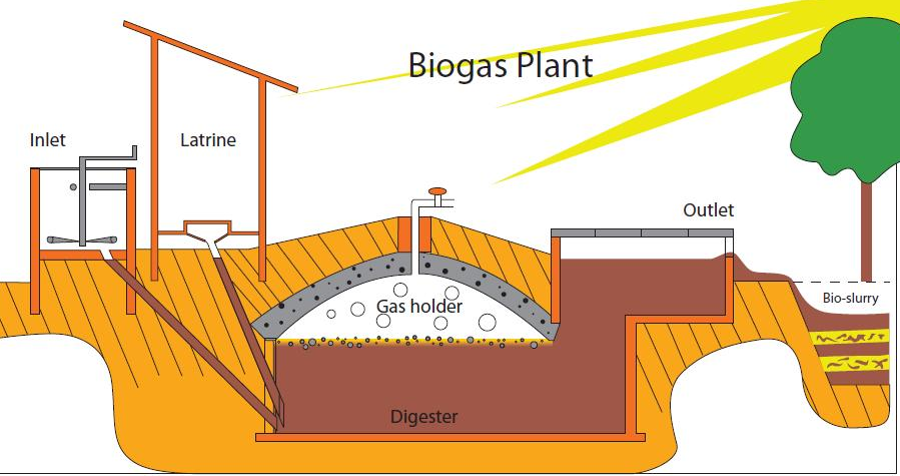
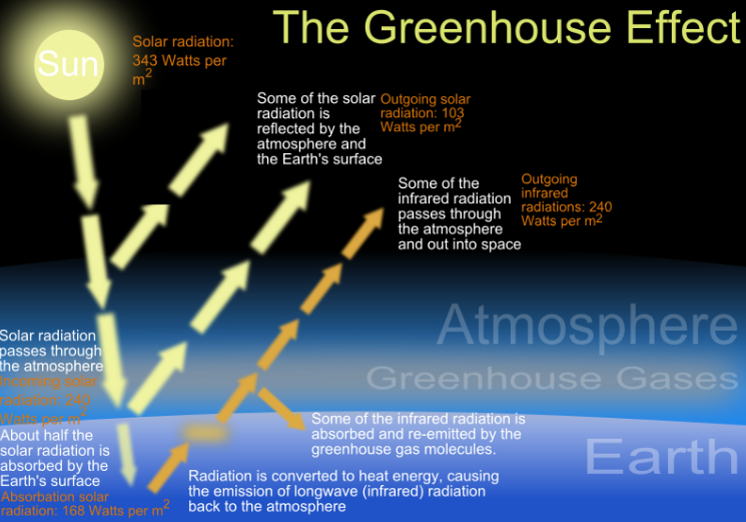
Energy transformations Conversion of energy from one form to another involves ‘losses’, usually in the form of thermal energy. Energy Efficiency = Useful energy output x100% Total energy input Energy transformations in a coal-fired power station •A number of energy transformations are involved in a coal-fired power station: •Coal is burnt- chemical energy from the coal is converted to thermal (heat) energy •The burning coal boils water: thermal energy (coal)--> thermal energy (steam). •The steam turns turbines- thermal energy ---->mechanical energy •Electricity is produced- mechanical energy -----> electrical energy Combustion reaction for the burning of coal C (s) + O2 (g) --> CO2 (g) This releases 32 KJ/g of energy. Electricity from natural gas •Energy from a gas-powered power plant is produced from burning methane and small alkanes. Thermal energy is converted to mechanical energy and then to electrical energy. Combustion reaction: CH4 (g) + 2O2 (g) --> CO2 (g) + 2 H2O (g) This releases 55.6 KJ/g of energy. Energy from biogas The combustion reaction also involves combustion of methane. The energy released here is less than that of natural gas - this is due to a lower methane content. Electricity from biogas is usually formed using small generators on site. Transport and fuels Fossil fuels are mainly used to power our transport vehicles. Fuels used are petroleum, diesel and LPG. These fuels add CO2 emissions to the atmosphere as well as other pollutants. Combustion reactions: •Petrol- octane combustion 2C8H18 (l) + 25 O2 (g) à 16 CO2 (g) + 18 H2O (l) Produces 47.8 KJ/g of energy •Liquid petroleum gas (LPG)- propane combustion C3H8 (l) + 5 O2 (g) à 3 CO2 (g) + 4 H2O (l) Produces 50.5 KJ/g of energy There is now a move to look at renewable fuels such as bioethanol. Bioethanol is produced from crops such as sugar cane. Bioethanol is renewable since the carbon dioxide produced is balanced out by the carbon dioxide absorbed by the sugar cane in photosynthesis. However, bioethanol is not carbon neutral as energy is used in transportation of the fuel and growing of the sugar cane. Combustion reaction: C2H5OH (l) + 3 O2 (g) à 2 CO2 (g) + 3 H2O (l) Produces 29.7 KJ/g of energy Environmental issues The green house effect occurs when green house gases such as carbon dioxide trap in heat. The more green house gas emissions the more heat is trapped- leading to global warming. Petrodiesel vs Biodiesel •Petrodiesel produces 10% more energy than Biodiesel, but biodiesel burns more smoothly and efficiently. •Biodiesel has a higher viscosity- does not flow as easily through fuel lines. Viscosity- the ease of flow of a fluid. High viscosity- slower flow. •At low temperatures biodiesel loses its ability to flow- it can even solidify. (Cloud point). Effects engine running. Cloud point- measure of performance at low temperature- the temperature at which small crystals start to form. Chemical structures: Petrodiesel- smaller non-polar molecules. -Weak dispersion forces between molecules Biodiesel- longer chain -Two highly electronegative oxygen atoms- polar -Dipole- dipole bonds - Polar bonds in biodiesel increase its viscosity and melting point. It also makes it more hygroscopic. Hygroscopic- a substance that absorbs water easily. -Lower storage time due to Biodegradation. Biodegradation- the breakdown of a substance by the action of living organisms- bacteria/fungi |
Coal-fired power plant diagram courtesy of By Tennessee Valley Authority - tva.com, Commons Archive, Public Domain, https://commons.wikimedia.org/w/index.php?curid=24393916
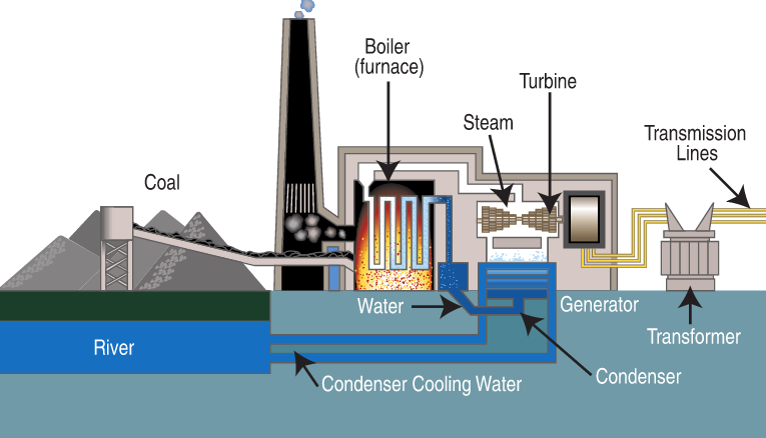
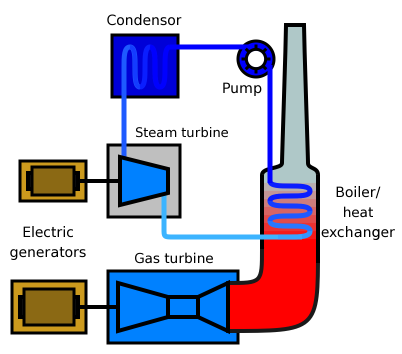
A gas-fired combined cycle power plant produces electricity from two types of turbine: gas and steam turbines.
Diagram courtesy of Public Domain, https://commons.wikimedia.org/w/index.php?curid=448105 Diagram courtesy of By SNV - SNV, Public Domain, https://commons.wikimedia.org/w/index.php?curid=8793880
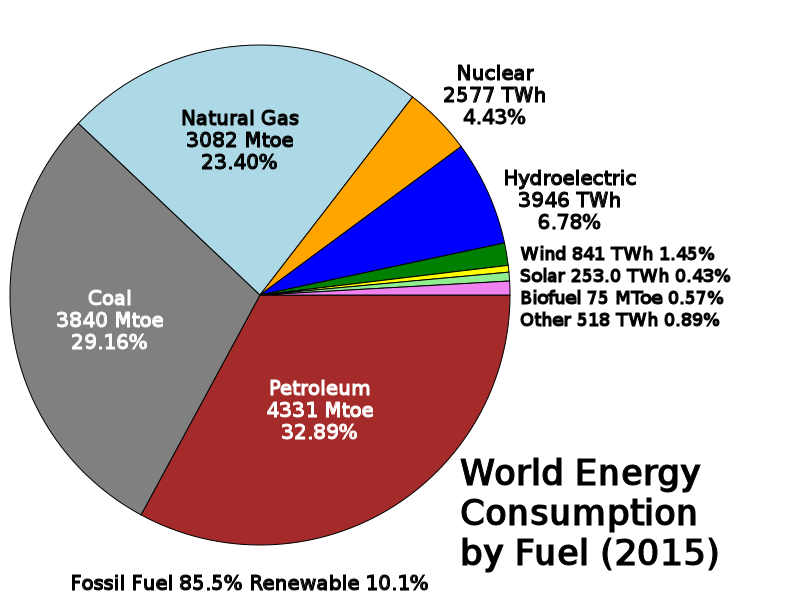
Diagram courtesy of By Delphi234 - Own work, CC0, https://commons.wikimedia.org/w/index.php?curid=33583618
The green house effect diagram courtesy of By ZooFari - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=5776439
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning