|
Functional groups
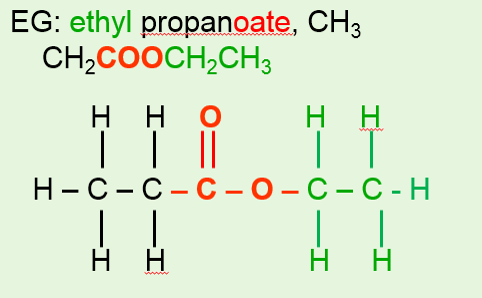
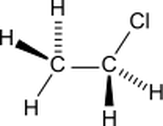
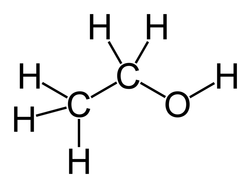
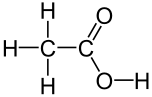
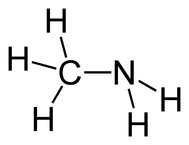
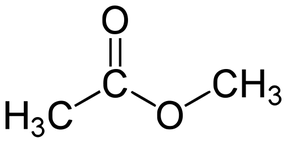
•The bond, atom, or group of atoms which gives a molecule its specific properties is called its functional group. •Eg: alkenes is C = C; alcohols is –OH. •More Homologous Series: 1.Haloalkanes (-X where X is a halogen). 2.Alcohols ( -OH) 3.Carboxylic acids (-COOH) 4.Amines (-NH2) 5.Esters ( -COO-) 1.Haloalkanes (-X where X is a halogen). •Halogen – Group VII elements eg: Cl, Br, I •Halogen replaces hydrogen atom. Example: chloromethane, CH3Cl (anaesthetic) 2. Alcohols (-OH). •Functional group is -OH •Alcohols names end in –ol. Number chain from the end of the closest carbon to the functional group. •Ethanol, CH3CH2OH present in wine, beer, etc •Example: methanol, CH3OH •Can be used as fuels, alcoholic drinks •Boiling points increase as the size of the molecule increases. •Polar –OH group allows alcohol to dissolve in water. However solubility decreases with longer chains. 3. Carboxylic Acids (-COOH). •Functional group is –COOH •Weak acids, relatively high boiling points •Carboxylic acids names end in –oic acid •Example: ethanoic acid, CH3COOH •The carboxyl carbon is always number 1. •Smaller molecules are soluble in water. 4. Amines (-NH2). •Functional group is -NH2 •Amines names end in –amine. •Uses: Found in many natural & synthetic drugs • Example: methanamine, CH3 NH2 5. Esters (-COO-). •Functional group is –COO- •Esters are formed by a reaction between a carboxylic acid and an alcohol. •Called an esterification reaction, also condensation reaction. Give fruit their pleasant ‘fruity’ smell, they have lower boiling points than alcohols and carboxylic acids of a similar size. Naming an ester 1.Locate the functional group and the carbonyl carbon. 2.Look at the C’s attached to the oxygen in the functional group- name using the prefixes (first part of name)- from alcohol 3.Look at the total no. of C’s attached to the other end of the functional group (including the carbonyl carbon)- name using the prefixes (second part of name)-from carboxylic acid. 4.Add the ending - OATE |
Pictures courtesy of wikimedia commons Chloroethane Ethanol
Ethanoic acid (Acetic acid) Methylamine Methyl ethanoate
Table of esters and their smells- https://jameskennedymonash.wordpress.com/2013/12/13/infographic-table-of-esters-and-their-smells/ |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning