Organic chemistry
•Compounds of carbon.
•Make up over 90% of all known chemicals.
•Why?
1. carbon-carbon bonds are strong.
2. carbon can bond to form long chain like structures and rings.
3. compounds that contain C-H bond are non polar, insoluble in water and do not react with water.
4. carbon can chemically bond to itself with single, double or triple bonds.
Source of hydrocarbons
•Fossil fuels:
1.coal
2. crude oil
3. natural gas.
•Fossil fuels are mainly chemical compounds called hydrocarbons (contain only carbon and hydrogen).
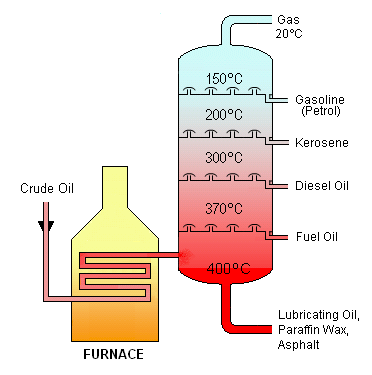
•Crude oil may be broken down intro fractions by a process called fractional distillation.
Diagram courtesy of Wikimedia commons.
Catalytic cracking
•Large molecules from fractional distillation of crude oil are broken into smaller molecules using temperature and catalysts in a process called cracking.
•Smaller molecules such as octane (used in motor fuel) and ethene (used for the manufacture of plastic) are made from cracking.
catalyst
•C10H22 (g) → C8H18 (g) + C2H4 (g)
decane octane ethene
Hydrocarbons
•Hydrocarbons – molecules that contain only carbon & hydrogen.
•Homologous series – any series of organic compounds in which each successive member differs by CH2 from the previous one.
•Homologous Series:
•Alkanes
•Alkenes
•Alkynes
•Cyclic Alkanes
•Compounds of carbon.
•Make up over 90% of all known chemicals.
•Why?
1. carbon-carbon bonds are strong.
2. carbon can bond to form long chain like structures and rings.
3. compounds that contain C-H bond are non polar, insoluble in water and do not react with water.
4. carbon can chemically bond to itself with single, double or triple bonds.
Source of hydrocarbons
•Fossil fuels:
1.coal
2. crude oil
3. natural gas.
•Fossil fuels are mainly chemical compounds called hydrocarbons (contain only carbon and hydrogen).
•Crude oil may be broken down intro fractions by a process called fractional distillation.
Diagram courtesy of Wikimedia commons.
Catalytic cracking
•Large molecules from fractional distillation of crude oil are broken into smaller molecules using temperature and catalysts in a process called cracking.
•Smaller molecules such as octane (used in motor fuel) and ethene (used for the manufacture of plastic) are made from cracking.
catalyst
•C10H22 (g) → C8H18 (g) + C2H4 (g)
decane octane ethene
Hydrocarbons
•Hydrocarbons – molecules that contain only carbon & hydrogen.
•Homologous series – any series of organic compounds in which each successive member differs by CH2 from the previous one.
•Homologous Series:
•Alkanes
•Alkenes
•Alkynes
•Cyclic Alkanes
