GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
Chemicals from Electrolysis in industry
|
To be viable electrolysis must:
—Be located in places with access to cheap and reliable electrical power. —Use electrolytes that are cheap and readily available - Use inexpensive electrodes - Be situtated near inexpensive transport —Consider the impact on the environment. Molten electrolytesElectrolysis using solution can reduce water at the cathode instead of the dersired metal. Thus electrolysis with molten salts is used instead.
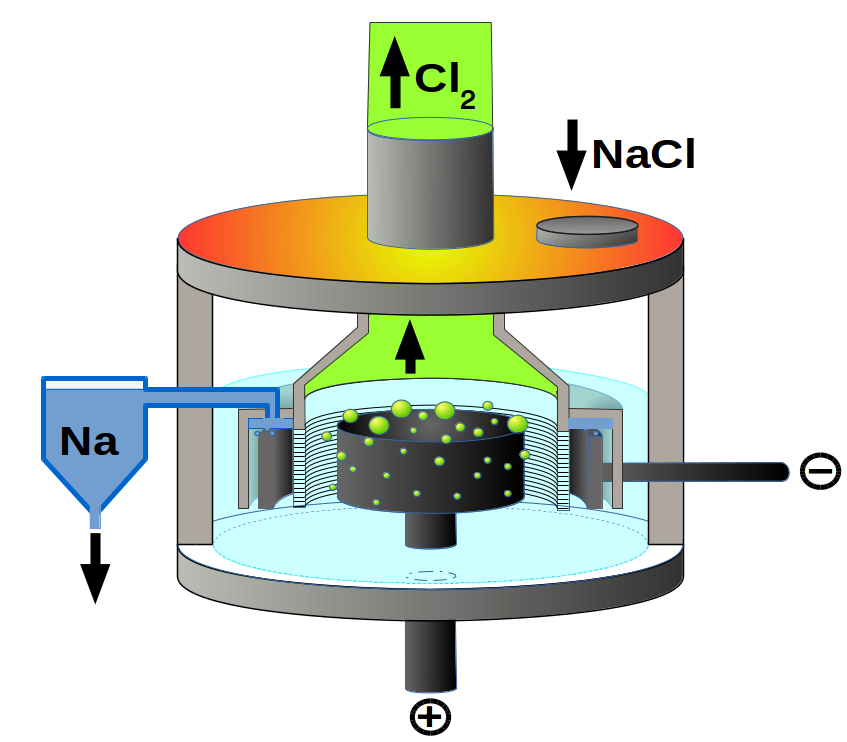
This is the case for producing sodium, potassium, calcium and aluminium metals. The use of a molten electrolyte usually involves greater energy expenditure and greater wear on the cell. In a molten electrolytic cell: Cations are reduced at the cathode (negative). Anions are oxidised at the anode (positive) Sodium •Sodium is extracted from molten sodium chloride by electrolysis (Downs cell).
•A simple cell of molten salt is shown on the right. The electrodes are made of an unreactive conducting material such as graphite or platinum. •The reaction occurring here is the reverse of the reaction that occurs spontaneously between sodium and chlorine to form sodium chloride. •Electrical energy from the power supply has caused a non-spontaneous reaction to occur •At the cathode (-) •The power supply pushes electrons towards this electrode. •Positive sodium ions accept electrons and become sodium atoms: Na+(l) + e- → Na(l) •Sodium is solid at room temp, but is liquid at the temperatures required to melt NaCl. •It is less dense than molten NaCl and floats to the top of the cell. •At the anode (+): •Electrons are withdrawn from this electrode by the power supply. •Since the electrode is made of unreactive material, the electrode itself will not participate in an oxidation reaction to supply electrons. •Instead Cl- ions in the electrolyte give up electrons and form chlorine atoms. •These atoms quickly form molecules of Cl2 and bubbles of chlorine gas form at the electrode. 2Cl-(l) → Cl2(g) + 2e- 2Na+(l) + 2Cl-(l) → 2Na(l) + Cl2(g) •Since Cl2 is a strong oxidant and Na is a strong reductant, there must be no contact between them; otherwise they will reform sodium chloride and the products from the electrolysis will be lost. •This will certainly occur in the simple cell. •In order to produce sodium and chlorine commercially, a modified cell, known as the Downs cell has been developed to minimise contact between the two products. A screen is used to prevent contact between the chlorine gas formed at the anode and the sodium formed at the cathode •Compared with cells using molten electrolyte such as used to produce sodium and aluminium, electrolytic cells using aqueous electrolytes tend to use less energy and have lower operating costs.
|
Downs cell diagram courtesy of PChemiker [CC BY-SA 4.0 (http://creativecommons.org/licenses/by-sa/4.0)], via Wikimedia Commons
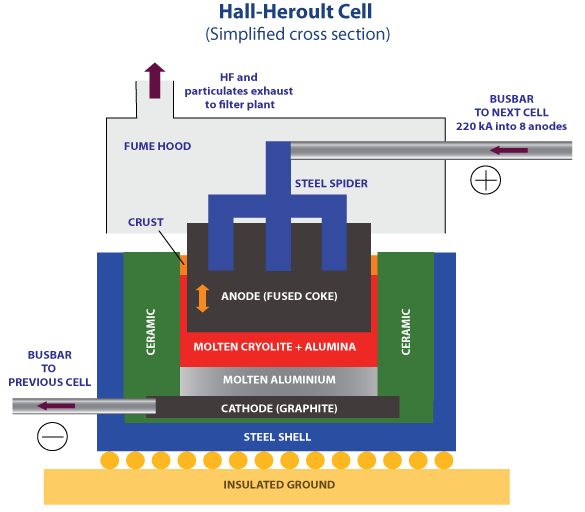
Hall- Heroult cell (Aluminium production) courtesy of Kashkhan at English Wikipedia, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=6864903
|