IONIC BONDING
Ionic bonding involves the transfer of electrons. If a substance loses electrons it becomes positively charged, if a substance gains electrons it becomes negatively charged.
Ionic bonding involves the transfer of electrons. If a substance loses electrons it becomes positively charged, if a substance gains electrons it becomes negatively charged.
iThe IONIC BOND is the result of very strong electrostatic attraction between two ions, in this case the attraction the attraction between the sodium and chloride ions.
This can be represented in the following way:
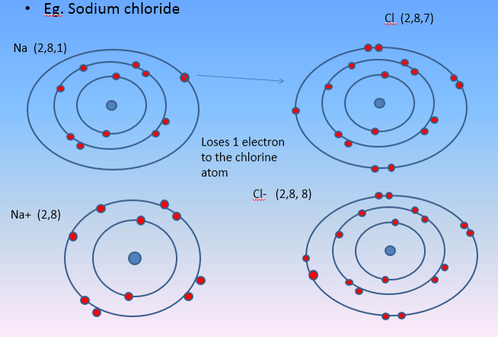
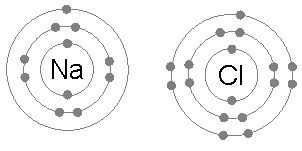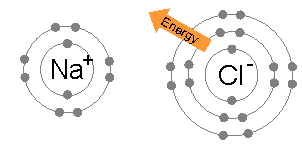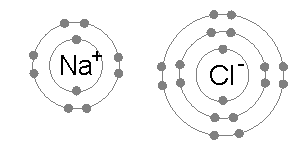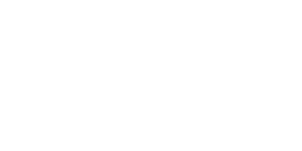
Na (2,8,1) + Cl (2,8,7) --> Na+ (2,8) + Cl- (2,8,8)
The Electrovalency (charge) that an ion carries relates to the number of electrons in its outer shell.
Eg. Group 1 have 1 electron in the outer shell which will be lost in bonding. Therefore, the valency will be +1 since one negative electron is lost.
Rules for naming ionic compounds
•The positive ion is written first (metal).
•Overall charge on the resulting compound is 0 (neutral).
•Subscripts are used to show ratios of ions.
•Brackets are used if more than one complex ion is contained in a compound.
Eg. Al2(SO4)3
•Cations with more than one valency are identified with the use of Roman Numerals.
Eg. Cu(I) , Cu(II)
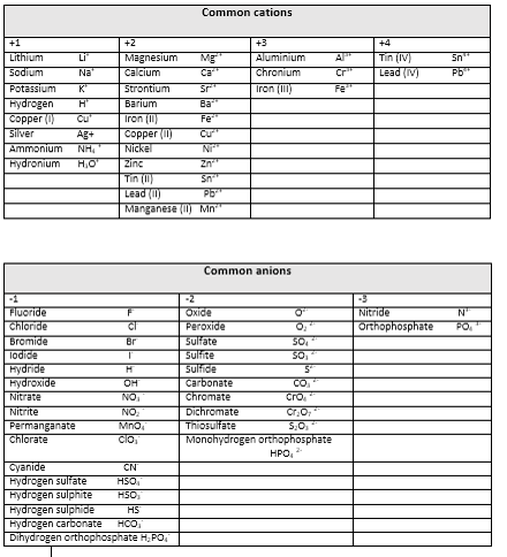
See Valency table below.
Examples:
1. Sodium chloride - ions Na+ Cl- each has a charge of 1, negative and positive cancel out. (So if the charges are equal, just put symbols together, drop the charges).
NaCl
2. Iron(III) oxide- ions Fe 3+ O2-
To balance the charges, you need to times Fe3+ by two this will give an overall positive charge of 6+ and O2- by three to give an overall negative charge of 6-.
2x Fe ^3+ 3x O^2- Fe^2O^3
3. Magnesium hydroxide- ions Mg 2+ OH-
To balance the charges, you need to times OH- by two this will give an overall negative charge of 2-.
Mg ^ 2+ 2x OH- Mg(OH)2
Note that hydroxide is made up from 2 elements, oxygen and hydrogen- both of these need to be doubled- therefore brackets need to be used.
Image- wikimedia commons
This can be represented in the following way:
Na (2,8,1) + Cl (2,8,7) --> Na+ (2,8) + Cl- (2,8,8)
The Electrovalency (charge) that an ion carries relates to the number of electrons in its outer shell.
Eg. Group 1 have 1 electron in the outer shell which will be lost in bonding. Therefore, the valency will be +1 since one negative electron is lost.
Rules for naming ionic compounds
•The positive ion is written first (metal).
•Overall charge on the resulting compound is 0 (neutral).
•Subscripts are used to show ratios of ions.
•Brackets are used if more than one complex ion is contained in a compound.
Eg. Al2(SO4)3
•Cations with more than one valency are identified with the use of Roman Numerals.
Eg. Cu(I) , Cu(II)
See Valency table below.
Examples:
1. Sodium chloride - ions Na+ Cl- each has a charge of 1, negative and positive cancel out. (So if the charges are equal, just put symbols together, drop the charges).
NaCl
2. Iron(III) oxide- ions Fe 3+ O2-
To balance the charges, you need to times Fe3+ by two this will give an overall positive charge of 6+ and O2- by three to give an overall negative charge of 6-.
2x Fe ^3+ 3x O^2- Fe^2O^3
3. Magnesium hydroxide- ions Mg 2+ OH-
To balance the charges, you need to times OH- by two this will give an overall negative charge of 2-.
Mg ^ 2+ 2x OH- Mg(OH)2
Note that hydroxide is made up from 2 elements, oxygen and hydrogen- both of these need to be doubled- therefore brackets need to be used.
Image- wikimedia commons