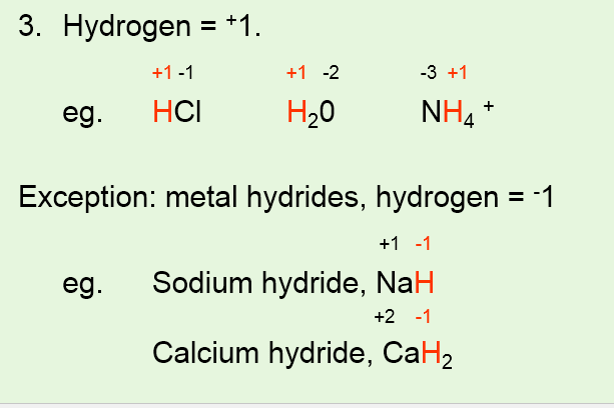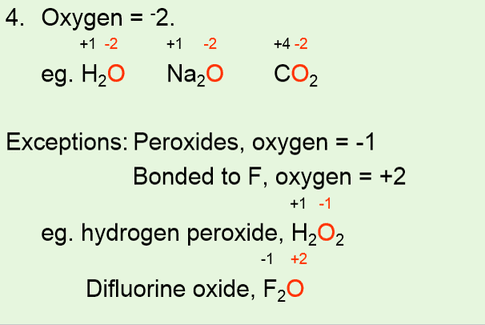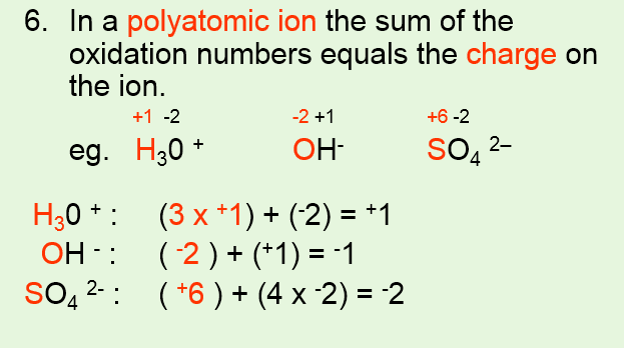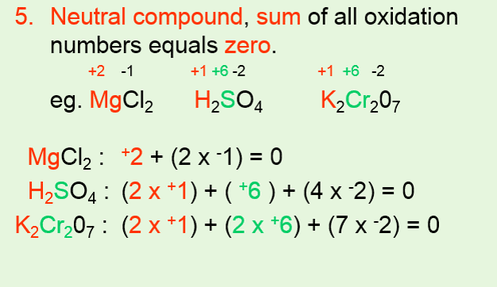STUDY CARD AVAILABLE- SEE CONTACT.
OXIDATION NUMBERS
•Not all redox reactions involve a transfer of electrons.
•It is possible for electrons to be shared rather than transferred.
•For Example:
C (s) + O2 (g) → CO2 (g)
and
2H2 (g) + O2 (g) → 2H20 (l)
•These reactions are classified as redox because they involve the oxidation of carbon and hydrogen and the reduction of oxygen.
•In both reactions electrons are not transferred but shared because the chemicals involved are molecules whose atoms are held together by covalent bonds.
•Oxidation numbers are allocated to each atom in any element or compound.
•Oxidation is an increase in the oxidation number of an atom.
•Reduction is a decrease in the oxidation number of an atom.
•Not all redox reactions involve a transfer of electrons.
•It is possible for electrons to be shared rather than transferred.
•For Example:
C (s) + O2 (g) → CO2 (g)
and
2H2 (g) + O2 (g) → 2H20 (l)
•These reactions are classified as redox because they involve the oxidation of carbon and hydrogen and the reduction of oxygen.
•In both reactions electrons are not transferred but shared because the chemicals involved are molecules whose atoms are held together by covalent bonds.
•Oxidation numbers are allocated to each atom in any element or compound.
•Oxidation is an increase in the oxidation number of an atom.
•Reduction is a decrease in the oxidation number of an atom.
|
IDENTIFYING REDOX REACTIONS •Using oxidation numbers to identify a redox reaction. •Use 6 rules to assign oxidation numbers to each atom of each reactant and product. •For a redox reaction to occur, one element needs to have an increase in oxidation number and another needs to have a decrease in oxidation number. |