Non polar covalent bonds
•Bonding electrons are shared equally and are uniformly distributed between nuclei of the 2 bonded atoms.
•Examples:
1. Hydrogen, H2
2. Oxygen, O2
3. Carbon Disulfide, CS2
(Note: Carbon and Sulfur have the same electronegativities).
Polar covalent bonds
•Bonding electrons are unequally shared and therefore unevenly distributed between nuclei of the 2 bonded atoms.
•Polar covalent bonds occur between atoms of different electronegativities.
•Examples:
1. Hydrogen Chloride, HCl
2. Carbon Dioxide, CO2 (bonds polar- but not a polar molecule)
Non Polar Molecule
•Molecules that contain only non polar bonds.
•Molecules which have polar bonds that cancel each other out because the molecule is symmetrical around a central atom.
•Eg: Iodine, I2; Methane,CH4;
Carbon dioxide, CO2
Polar Molecules
•Polar molecules are molecules that have:
1. Polar covalent bonds.
2. The molecule is not symmetrical around a central atom.
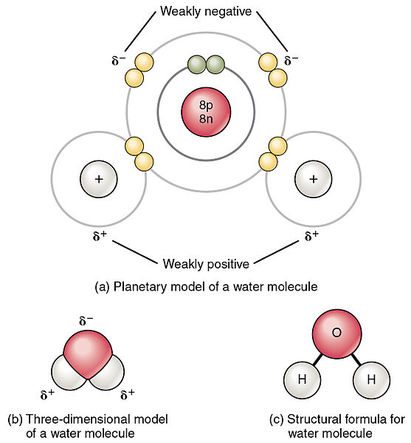
•Polar molecules are called dipolar.
Eg. Water
Diagram wikimedia commons
•Bonding electrons are shared equally and are uniformly distributed between nuclei of the 2 bonded atoms.
•Examples:
1. Hydrogen, H2
2. Oxygen, O2
3. Carbon Disulfide, CS2
(Note: Carbon and Sulfur have the same electronegativities).
Polar covalent bonds
•Bonding electrons are unequally shared and therefore unevenly distributed between nuclei of the 2 bonded atoms.
•Polar covalent bonds occur between atoms of different electronegativities.
•Examples:
1. Hydrogen Chloride, HCl
2. Carbon Dioxide, CO2 (bonds polar- but not a polar molecule)
Non Polar Molecule
•Molecules that contain only non polar bonds.
•Molecules which have polar bonds that cancel each other out because the molecule is symmetrical around a central atom.
•Eg: Iodine, I2; Methane,CH4;
Carbon dioxide, CO2
Polar Molecules
•Polar molecules are molecules that have:
1. Polar covalent bonds.
2. The molecule is not symmetrical around a central atom.
•Polar molecules are called dipolar.
Eg. Water
Diagram wikimedia commons