|
RATE AND EQUILIBRIUM REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-unit-3-and-4-rate-and-equilibrium-revision-11987322
FACTORS THAT AFFECT REACTION RATE There are five main ways that the rate of reaction can be increased: •Temperature •Catalysts •Concentrations of reactants •Surface area of a solid reactant •Pressure of gaseous reactants or products HOW DOES THE SURFACE AREA AFFECT THE RATE OF REACTION? If one of the reactants is a solid, the surface area of the solid will affect how fast the reaction goes. The two types of molecule can only bump into each other on the surface of the solid. So the larger the surface area of the solid, the faster the reaction will be. Smaller particles have a bigger surface area than larger particles for the same mass of solid- crushing a solid will increase the surface area. HOW DOES CONCENTRATION AFFECT THE RATE OF REACTION? •Increasing the concentration of the reactants will increase the frequency of collisions between the two reactants. More reactants present the more likely a successful collision. THE EFFECT OF PRESSURE ON TWO GASES The atoms or molecules in a gas are very spread out. For the two chemicals to react, there must be collisions between their molecules. By increasing the pressure, you squeeze the molecules together so you will increase the frequency of collisions between them. T |
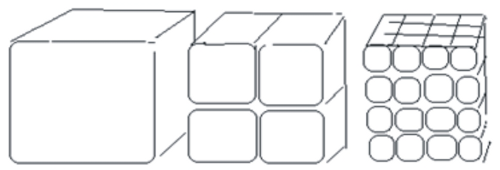
Change in surface area- smaller pieces give a larger surface area.
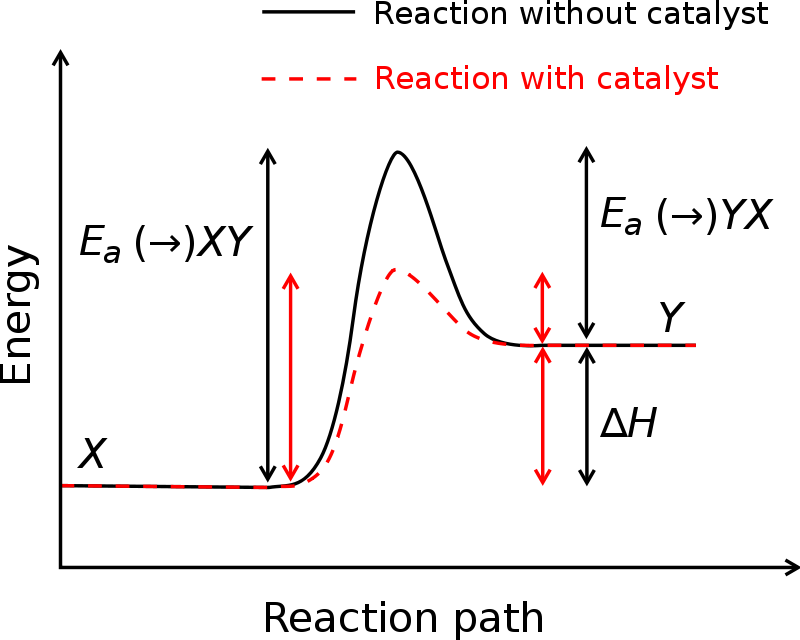
Diagram courtesy of By Julie Sandeen - http://cafreetextbooks.ck12.org/science/CK12_Earth_Science_rev.pdf (page 322)If the above link no longer works, visit http://www.ck12.org and search for CK-12 Earth Science., CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=10828845 HOW DOES TEMPERATURE AFFECT THE RATE OF REACTION? When two chemicals react, their molecules have to collide with each other with sufficient energy for the reaction to take place. The two molecules will only react if they have enough energy. By heating the mixture, you will raise the energy levels of the molecules involved in the reaction. Increasing temperature means the molecules move faster. This is kinetic theory. EXTENDING COLLISION THEORY •The effect of temperature on reaction rate cannot simply be explained by the increased frequency of collisions. An increase in temperature increases the kinetic energy of particles and thus the likelyhood of successful collisions. •A reaction can take place only if the particles colliding have more energy than the activation energy of the reaction. •At any instant only a fraction of the particles present have sufficient energy to participate in successful collisions and react. (Increasing the temperature, increases the energy and more particles have energy equal to or greater than the activation energy). CATALYSTS A catalyst is a chemical that will speed up a reaction. The catalyst does not react in the reaction itself). A calalyst provides a different pathway for the reaction to procede by lowering the activation energy needed for the reaction. A catalyst can be in the same state (homogeneous) or different state (heterogenous) as the products and reactants. Image courtesy of Copyrighted free use, https://commons.wikimedia.org/w/index.php?curid=779552
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning