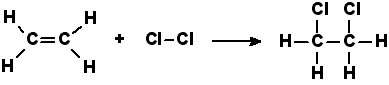ORGANIC CHEMISTRY REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-revision-organic-chemistry-12042644
Reactions of alkanes
|
•The alkanes are insoluble in water but are soluble in non-polar solvents.
•There are weak dispersion forces between molecules as evidenced by their low melting and boiling points. •Most reactions involving alkanes are either combustion or substitution. •Combustion: Fuel + Oxyen --> Carbon dioxide and water •Eg: CH4 (g) + 2O2 (g) → CO2 (g) + 2H20 (g) + energy methane •Substitution: Chlorine atoms take the place of one or more of the hydrogen atoms. •Eg: CH4 (g) + Cl2 (g) → CH3Cl (g) + HCl (g) methane chloromethane |
Methane combustion diagram By Jynto Robert A. Rohde Jacek FH Jynto [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
Methane substitution diagram V8rik at English Wikipedia [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0) or GFDL (http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons
|
Reactions of alkenes
|
Ethene is the first member of the Alkenes homologous series.
Properties of Ethene (C2H4): •Is unsaturated •Is a non-polar molecule •Is insoluble in water •Is a flammable gas •Participates in addition reactions •Polymerises to produce polyethene. •Combustion: Fuel + Oxygen → Carbon Dioxide + Water •Eg: C2H4 (g) + 3O2 (g) → 2CO2 (g) + 2H20 (g) ethene •Addition: C=C double bond is broken and one atom is added to each carbon atom to form a molecule that has only C-C single bonds. •Eg: C2H4 (g) + Cl2 (g) → C2H4Cl2 (g) ethene 1,2 – dichloroethane •Ethene also reacts with H2O to form an alcohol. H3PO4 is used as a catalyst along with a 300oC temperature to speed up the reaction. Reaction with water is called hydrolysis. Addition Polymerisation •A polymer is made up of a repeating unit or monomer. Each monomer is added onto the end of the chain – hence the name: addition polymerisation. •Monomers must contain a double bond. The double bond breaks to form a single bond and the free electrons form new bonds linking the monomer onto the chain. PolymerisationA polymer made from more than one monomer is called a copolymer.
e.g. Polyester •Polyester is a copolymer made from di-alcohol and di-carboxylic acid monomers. •Condensation reactions form water as a product •The alcohol has two hydroxy groups and the carboxylic acid has two carboxy groups. These monomers combine in a condensation reaction to form polyester chains. •Polyester has many uses in packaging and can be blow moulded to make soft-drink bottles. Some polyesters can be cross-linked, making them suitable for use as surface coatings, including paints. Haloalkanes.Haloalkanes (-X where X is a halogen).
•Halogen – Group VII elements eg: Cl, Br, I •Halogen replaces hydrogen atom. •Example: chloromethane, CH3Cl (anaesthetic) Chloroalkanes •Chlorine is highly electronegative- making the chloroalkane molecule polar. Chlorine readily reacts with other polar molecules like water, hydroxide ions and ammonia in a substitution reaction. |
Ethene addition reaction diagram Wikimedia commons.
|