STUDY CARD AVAILABLE- SEE CONTACT.
.REACTIVITY OF METALS
•When magnesium is placed in copper sulfate solution a redox reaction occurs, causing copper metal to form on the magnesium and the deep blue colour of the solution pales.
•The equation is:
Mg (s) + CuSO4 (aq) → Cu (s) + MgSO4 (aq)
•If you place a copper strip in a solution of magnesium sulfate, no reaction occurs.
•Therefore, it is possible to conclude that magnesium is more reactive than copper.
•Experience tells us that different metals have varying abilities to react with other substances.
•For example, potassium is so reactive that it is found naturally as a compound rather than a pure metal.
•Gold, on the other hand, is very unreactive. It occurs in nature as a pure element.
Potassium and gold are at the opposite of the reactivity series of metals (also called the activity series).
•Example: Reaction between magnesium metal and copper sulfate solution.
•Balanced Equation:
Mg (s) + CuSO4 (aq) → Cu (s) + MgSO4 (aq)
•Ionic Equation: (SO4 2- spectator ions)
Mg (s) + Cu 2+ (aq) → Cu (s) + Mg 2+ (aq)
•When magnesium is placed in copper sulfate solution a redox reaction occurs, causing copper metal to form on the magnesium and the deep blue colour of the solution pales.
•The equation is:
Mg (s) + CuSO4 (aq) → Cu (s) + MgSO4 (aq)
•If you place a copper strip in a solution of magnesium sulfate, no reaction occurs.
•Therefore, it is possible to conclude that magnesium is more reactive than copper.
•Experience tells us that different metals have varying abilities to react with other substances.
•For example, potassium is so reactive that it is found naturally as a compound rather than a pure metal.
•Gold, on the other hand, is very unreactive. It occurs in nature as a pure element.
Potassium and gold are at the opposite of the reactivity series of metals (also called the activity series).
•Example: Reaction between magnesium metal and copper sulfate solution.
•Balanced Equation:
Mg (s) + CuSO4 (aq) → Cu (s) + MgSO4 (aq)
•Ionic Equation: (SO4 2- spectator ions)
Mg (s) + Cu 2+ (aq) → Cu (s) + Mg 2+ (aq)
Half equations
•Half equations are a useful way of understanding the processes involved in a redox reaction.
•Although the oxidation and reduction reactions occur simultaneously, it is possible to write the two reactions separately.
•The two reactants (oxidant and reductant) have their own half equations.
•Example: Reaction between magnesium metal and copper sulfate solution.
•Ionic Equation: (SO4 2- spectator ions)
Mg (s) + Cu 2+ (aq) → Cu (s) + Mg 2+ (aq)
•Oxidation Half Equation:
Mg (s) → Mg 2+ (aq) + 2e-
•Reduction Half Equation:
Cu 2+ (aq) + 2e- → Cu (s)
•A conjugate redox pair is made up of two species (an oxidant and a reductant) which differ by the number of electrons that have been transferred from the oxidant to the reductant in the chemical reaction.
•Conjugate Redox Pairs are written as:
oxidant / reductant
•For Example: Mg 2+ / Mg; Cu 2+ / Cu
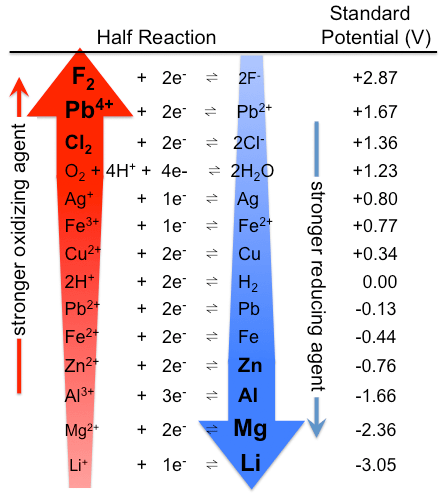
•The Electrochemical Series of Metals ranks the metals in order of their reactivity.
•The most reactive metals appear at the bottom of the table since they lose electrons more easily.
•The most reactive metals are the strongest reductants.
•The half equations are written in the direction in which reduction occurs.
•A double arrow (↔ ) is used to indicate that the reaction can proceed in both directions.
•The oxidants are on the left.
•The reductants are on the right.
•The strongest oxidants are top left of the table.
Diagram of electrochemical series courtesy of By Tem5psu (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
Using Electrochemical series to predict redox reactions.
•To have a redox reaction you need an oxidant (left side of table) and a reductant (right side of table).
•To have a spontaneous reaction, the oxidant must be above the reductant on the table.
•Example 1: Sodium metal (Na) added to a lead nitrate solution (Pb(NO3)2).
1.Look at Electrochemical Series.
2.Pb 2+ is the oxidant, Na is the reductant.
3.Pb 2+ is above Na on the table.
4.A spontaneous redox reaction will occur.
5.Ionic Equation:
2Na (s) + Pb 2+ (aq) → Pb (s) + 2Na + (aq)
reductant oxidant reductant oxidant
•Example 2: Tin metal (Sn) added to a aluminium nitrate solution (Al(NO3)3).
1.Look at Electrochemical Series.
2.Al 3+ is the oxidant, Sn is the reductant.
3.Al 3+ is below Sn on the table.
4.A spontaneous redox reaction will not occur (no reaction).
•To have a redox reaction you need an oxidant (left side of table) and a reductant (right side of table).
•To have a spontaneous reaction, the oxidant must be above the reductant on the table.
•Example 1: Sodium metal (Na) added to a lead nitrate solution (Pb(NO3)2).
1.Look at Electrochemical Series.
2.Pb 2+ is the oxidant, Na is the reductant.
3.Pb 2+ is above Na on the table.
4.A spontaneous redox reaction will occur.
5.Ionic Equation:
2Na (s) + Pb 2+ (aq) → Pb (s) + 2Na + (aq)
reductant oxidant reductant oxidant
•Example 2: Tin metal (Sn) added to a aluminium nitrate solution (Al(NO3)3).
1.Look at Electrochemical Series.
2.Al 3+ is the oxidant, Sn is the reductant.
3.Al 3+ is below Sn on the table.
4.A spontaneous redox reaction will not occur (no reaction).