|
STUDY CARD AVAILABLE- SEE CONTACT.
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646 THE ELECTROCHEMICAL SERIES
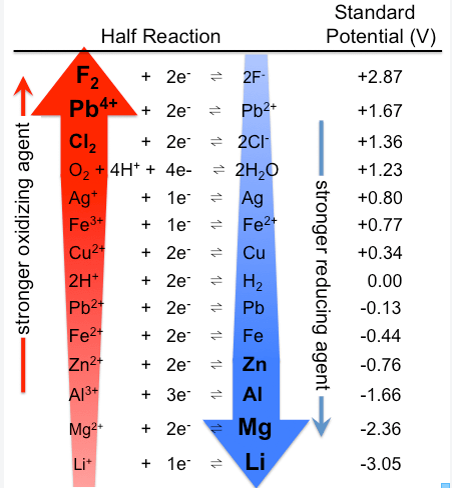
•The electrochemical series is valid only for the conditions under which it was determined. •The series that you will be supplied with applies at a temperature of 25°C, a pressure of 1 atm and 1M concentration of solutions. •When reading the electrochemical series, the higher one is written forwards and undergoes reductions, while the lower one is written in reverse and undergoes oxidation. POTENTIAL DIFFERENCE •A current flows due to the difference of electron pushing power in each of the half cells. Thus there is a potential difference between the two half cells. •The potential difference is sometimes called the emf. •The potential difference is measured in volts and measures the amount of energy supplied by a fixed amount of charge flowing from a galvanic cell. •An indication of the cell voltage can be found using the electrochemical series. •Potential differences of cells at standard conditions can be found using the E° values in the electrochemical series. •These standard half cell potentials give a numerical measure of the tendency of a half cell reaction to occur as a reduction reaction. •E° values are measured by connecting two half cells to a reference cell. •A hydrogen half cell is used for this purpose and its E° is assigned as a 0. •The potential difference of a cell at standard conditions is the difference between the E° values of its two half cells. Cell potential difference = higher half cell E° - lower half cell E° PREDICTING SPONTANEOUS REDOX REACTIONS
•In a galvanic cell, the higher half reaction in the electrochemical series undergoes reduction in the forward direction and the lower reaction undergoes oxidation in the reverse. •This principle applies equally to redox reactions that occur when reactants are mixed directly. LIMITATIONS •The standard half cell potentials are measured under standard conditions - so figures change with differing conditions. •The electrochemical series also does not give an indication of the rate of reactions. |
Diagram of electrochemical series courtesy of By Tem5psu (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
FINDING THE POTENTIAL DIFFERENCE
Example: •Two half cell reactions, determine the overall equation along with the potential difference. • Cu+ (aq) + e- --> Cu (s) + 0.52 V Fe2+ (aq) + 2e- --> Fe (s) - 0.44 V •The copper equation appears higher in the table- so this equation stays the same (Stronger oxidant) •The Iron equation is lower in the table, so reverse equation – this will change the voltage to a positive. •Make sure equation is balanced by balancing the electrons on either side. •Overall equation: 2Cu+ (aq) + Fe (s) --> 2Cu (s) + Fe2+ (aq) E0 = + 0.52 - - 0.44 or +0.52 + 0.44 = +0.96V Reaction is Spontaneous if the Oxidant reaction is higher in the table – gives a positive EMF. |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning