|
WHAT ARE POLYMERS?
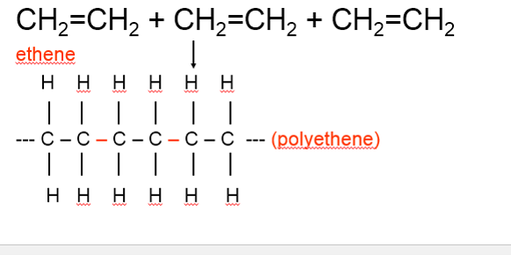
•A polymer molecule is made up of a large number of units called monomers joined together in chains. •When monomers link together to form a polymer chain the process is called polymerisation. •Copolymers are formed when two or more different monomers are used. THERMOSOFTENING POLYMERS •Plastics that may be repeatedly melted, reshaped or hardened by cooling are called thermosoftening polymers or thermoplastics. •Examples: 1.Polyvinyl chloride (PVC) 2.Nylon 3.Polystyrene 4.Polytetrafluoroethylene (teflon) 5.Polymethyl methacrylate (perspex) THERMOSETTING POLYMERS •Plastics which do not melt but char when heated are called thermosetting polymers. These plastics must be moulded or shaped during their manufacture. •Examples: 1.Urea Formaldehyde (Bakelite) 2.Epoxy Resin (Araldite) 3.Melamine Formaldehyde (Formica) 4.Polyurethane ADDITION POLYMERISATION •Ethene undergoes an addition reaction (double bond in alkene is broken) with itself to form the polymer, polyethene. •The length of the polymer chains can vary from 100 to 1000 carbon atoms. •Modifying ethene by substituting different functional groups produces other monomers which make polymers with different properties. •Examples: polyethene, polyvinyl chloride (PVC), polystyrene, teflon. |
Image courtesy of By Nolabob - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=37065954
|
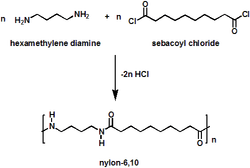
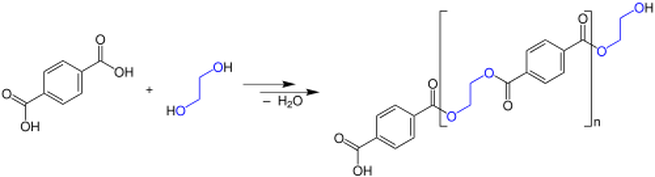
CONDENSATION POLYMERISATION
•Condensation polymers are formed by the “head to tail” joining of monomer units.
•Each join (link) is accompanied by loss of a small molecule such as water, H20.
•The reaction requires the presence of 2 reactive functional groups on each monomer.
Examples: nylon, polyester, cotton, wool, silk, protein, starch
Diagram- Making polyester- courtesy of wikimedia commons.