|
STUDY CARD AVAILABLE- SEE CONTACT.
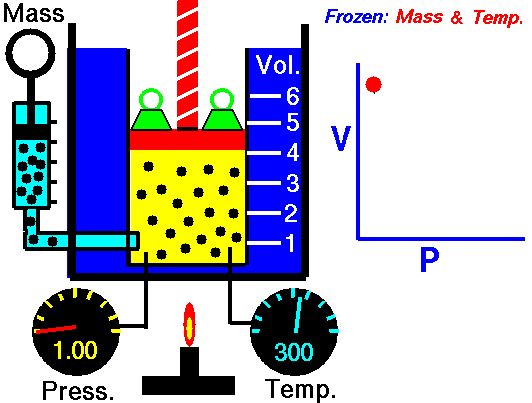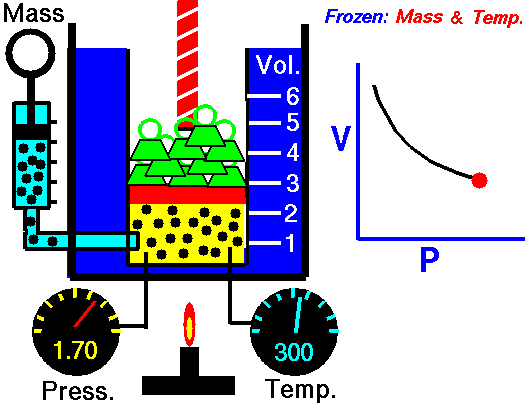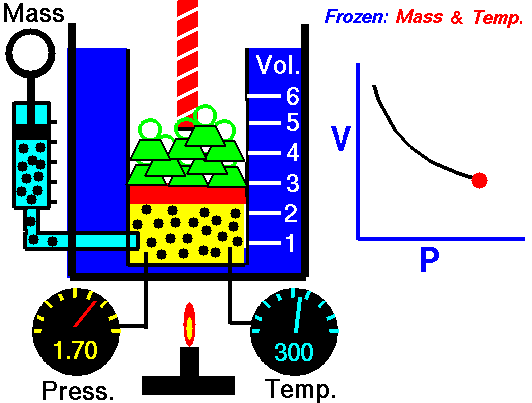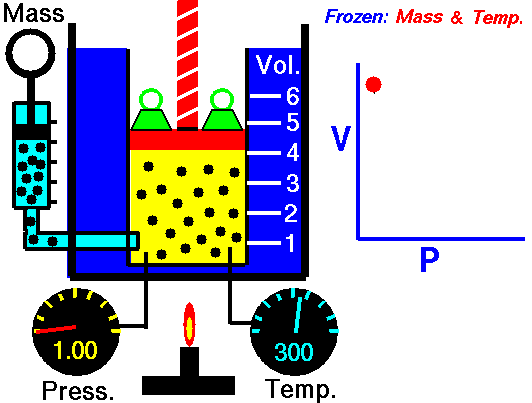
Relationship between Pressure- Volume (Boyle's Law)
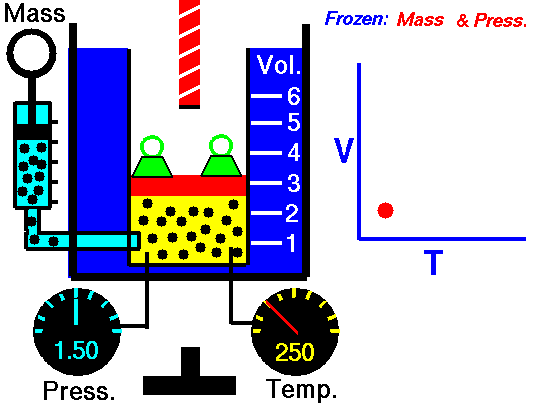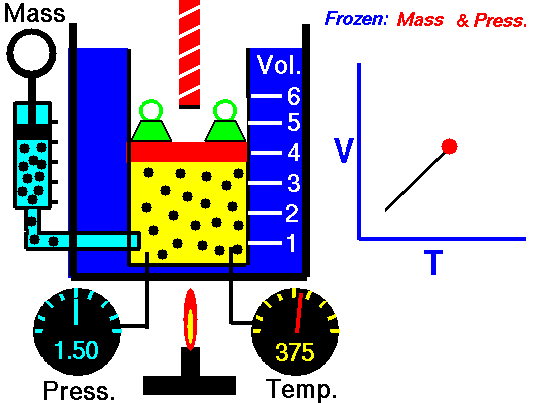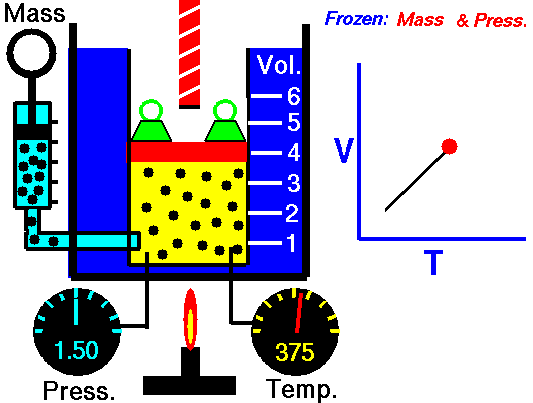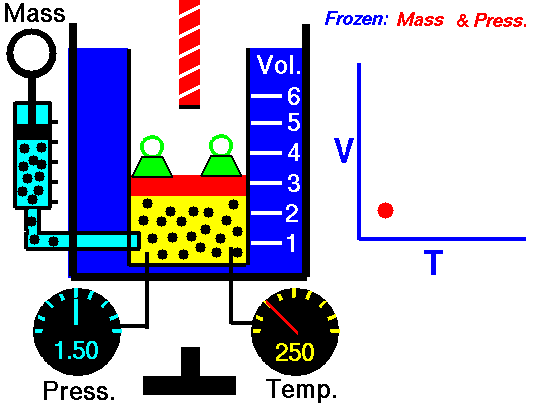
•The volume of a fixed mass of gas at constant temperature is inversely proportional to the pressure exerted on it. •P ∞ 1 / V then PV equals a constant value. •Boyle’s Law: P1V1 = P2V2 at constant temperature. Relationship between Temperature- Volume (Charles' Law) •The volume of a fixed mass of gas at constant pressure is directly proportional to its absolute temperature (kelvin scale). •V ∞ T then V / T equals a constant value. •Charles’ Law: V1 = V2 at constant pressure T1 T2 Relationship between Volume-Mole (Avogadro's Law) Avogadro postulated that equal volumes of gases at the same temperature and pressure contain equal numbers of particles. V ∞ n or V= kn where k is a constant. Molar gas Volume •Molar Gas Volume is the volume of 1 mole of a particular gas. •It has been found experimentally that 1 mole of any gas at STP occupies a volume of 22.7 L. •The molar volume of a gas at SLC is 24.8 L. •Gas constants: VSTP = 22.7 L VSLC = 24.8 L STP- 0 oC and 100kPa SLC – 25 oC and 100KPa There is a direct relationship between the number of moles of a gas and its molar volume. n = V Vm Where: n is the number of moles of gas V is the actual volume of gas Vm is the molar volume of the gas •Combine Boyle’s and Charles’ Laws: PV equals a constant value V / T equals a constant value to get PV / T equals a constant value. •Combined Gas Law: P1V1 = P2V2 for a constant amount of gas. T1 T2 •P ∞ 1 / V (Boyle’s Law) •P ∞ T (Gay Lussac’s Law) •P ∞ n •Combine these proportionalities: P ∞ nT / V Universal gas equation •Replace proportionality with equality sign. •Introduce a general gas constant, R and we get the general gas equation. PV = nRT where: Pressure (P) is measured in kilopascals (kPa) Volume (V) is measured in litres (L) Temperature (T) is measured in kelvin (K) Number of moles (n) – unit (mol) General Gas Constant (R) = 8.31JK-1mol-1 |
Boyle's Law animation courtesy of By NASA's Glenn Research Center - http://www.grc.nasa.gov/WWW/K-12/airplane/aboyle.html, Public Domain, https://commons.wikimedia.org/w/index.php?curid=4448761
Charle's Law animation courtesy of By NASA's Glenn Research Center - http://www.grc.nasa.gov/WWW/K-12/airplane/aglussac.html, Public Domain, https://commons.wikimedia.org/w/index.php?curid=4448734
Sample Problem using the universal gas equation Find the volume of 6.30 mol of carbon dioxide gas at 23oC and 550 kPa pressure. (R = 8.31 JK-1mol-1) Solution: Step 1: Write down given data in correct units. P = 550 kPa V = ??? n = 6.30 mol T = 23oC = 296 K R = 8.31 JK-1mol-1 Step 2: Use general gas equation to calculate volume. PV = nRT V = nRT / P = 6.30 x 8.31 x 296 / 550 = 28.2 L |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning