|
VOLUMETRIC ANALYSIS
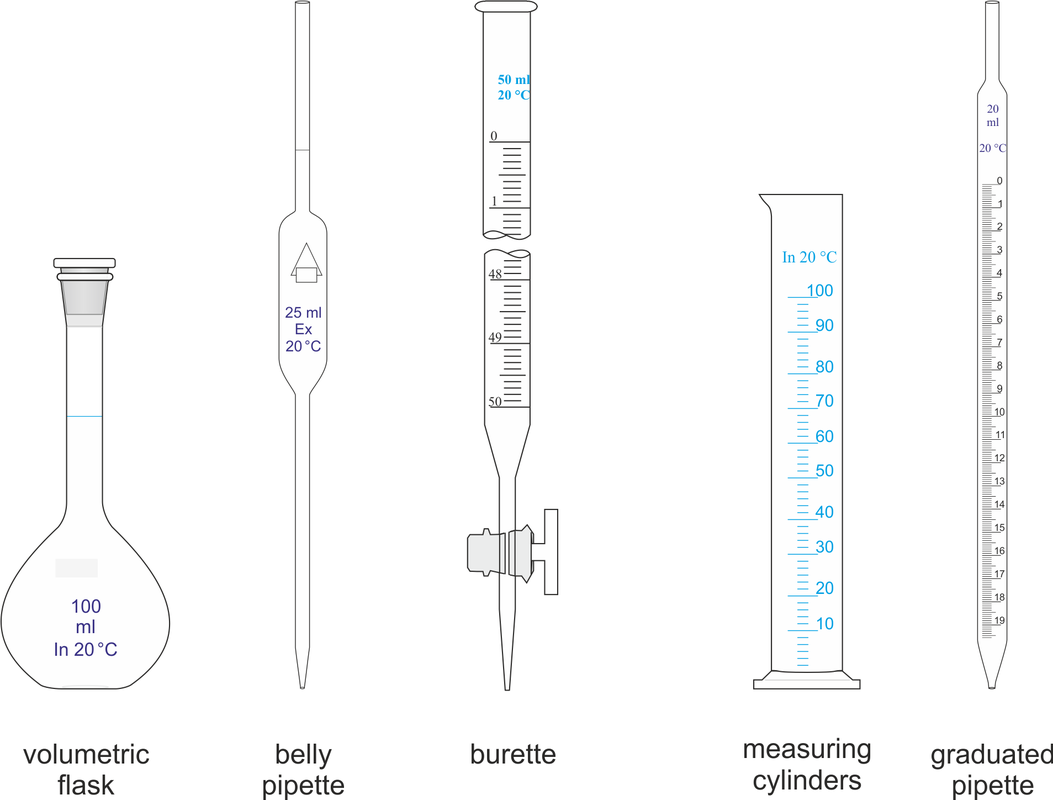
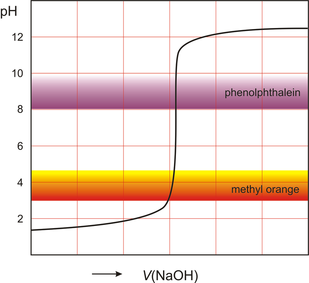
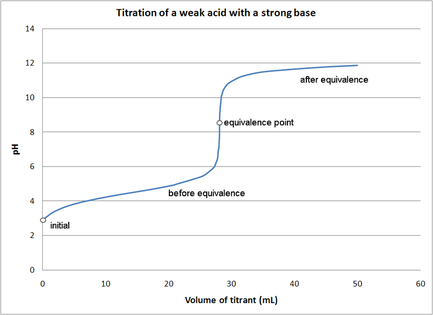
•A common way of finding an unknown concentration is by using the technique of Volumetric Analysis. This technique involves reacting a measured volume of a standard solution with a measured volume of the solution of unknown concentration. Steps involved in Volumetric Analysis: •pipette a known volume (aliquot) of solution into a conical flask •dispense other solution slowly through a burette •stop when indicator just changes colour and measure volume (titre) dispensed •repeat until you have three concordant titre results (no more than 0.1mL difference between all three) then average the concordant results to minimise errors. VOLUMETRIC ANALYSIS TERMS •Equivalence point-when chemically equivalent amounts of acid and base, according to mole ratio of the equation, are present. •End point-when the solution colour changes. •Titre- volume of solution added from the burette. •Aliquot- known volume of solution delivered from a pipette •Concordant titres- are those in a specified range eg. +/- 0.1ml of each other. •Pipette- apparatus used for delivering a known volume of solution from volumetric flask into conical flask •Burette – apparatus into which the unknown solution is placed. •Accuracy of Instruments used •Burette - within +/- 0.02mL •20 mL Pipette - within +/- 0.03mL •250mL volumetric flask – within +/- 0.3mL INDICATORS •An indicator is used during acid–base titration to identify the equivalence point of the reaction. An acid–base indicator is a substance whose colour depends on the concentration of H3O+ ions in solution. Indicators are weak acids with their acid form being one colour and their conjugate base being another. •The indicator must be chosen carefully to ensure that the point during the titration where the indicator changes colour, the end point, closely matches the equivalence point of the reaction. •If we follow the pH changes that take place during the titration of a solution of a strong base, such as a 0.1 M solution of sodium hydroxide, with a solution of a strong acid, such as 0.1 M hydrochloric acid, we would see a plot similar to that shown in Figure on the right. •At the end point, addition of a very small volume of strong base produces a large change in pH. This is referred to as a sharp end point. In the case of the reaction between a strong acid and a strong base the equivalence point falls close to pH 7. •When a weak base is titrated with a strong acid, or a strong base is titrated with a weak acid, there is a much more gradual change of pH around the end point (Figure 2 on the right). • In this situation, only indicators whose colour changes within the end point range can be used. •In the case of reactions between weak acids with weak bases the pH change is so gradual that this combination cannot be analysed by a simple, direct titration. Instead we use instruments such as a pH meter to find the equivalence point. Sample Titration Problem: •1.06g of anhydrous sodium carbonate is placed in a 250 ml volumetric flask and water added to make a standard solution. •This standard solution is titrated against a solution of hydrochloric acid. •It was found that a 20 ml aliquot of sodium carbonate solution required a titre of 22 ml of hydrochloric acid for neutralisation. •What is the concentration of the HCl? Solution: Step 1: Write a balanced equation. and identify known and unknown. Na2CO3(aq) + 2HCl(aq) → 2NaCl(aq)+ CO2 (g) )+ H20 (l) Known unknown 1.06 g C = ? Step 2: Calculate n (known). n(Na2CO3) 250 ml volumetric flask = m / M = 1.06 / (23 x 2 + 12 + 3 x 16) = 1.06 / 106 = 0.01 mol Calculate n (known) in 20ml. n(Na2CO3) 20 ml aliquot = 20 / 250 x 0.01 = 0.0008 mol Note 20ml aliquot divided by 250ml dilution Step 3: From equation find ratio of n(unknown) to n(known). n(HCl) = 2/1 x n(Na2CO3) 20 ml aliquot = 2/1 x 0.0008 = 0.0016 mol Step 4: Answer Question Find concentration of HCl. V (HCl) = 22 ml = 0.022 L C (HCl) = n / V = 0.0016 / 0.022 = 0.07 M Precision and errors •Precision- when repeated measurements are close to each other. •Accuracy- when measurements are close to the true value. Types of errors- •Mistakes- eg. human error in reading scales •Systematic errors- when equipment is out- scales incorrect etc., incorrect indicators used. •Random errors- follow no regular pattern. |
Volumetric glassware image courtesy of E. Generalic, http://www.periodni.com/enig/volumetrijske_metode_analize.html
Titration of a strong acid adding a strong base.
Diagram courtesy of E. Generalic, http://glossary.periodni.com/glossary.php?en=acid-base+titration Titration of a weak acid with a strong base.
Diagram courtesy of By Quantumkinetics - Own work, CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=14947161 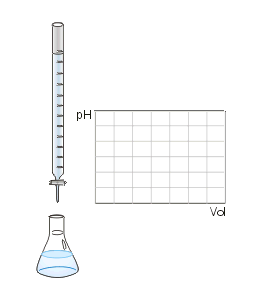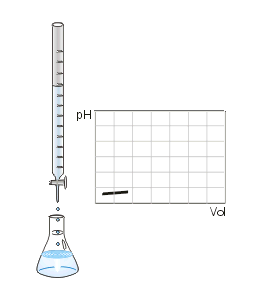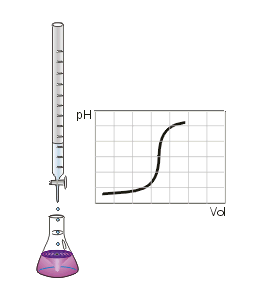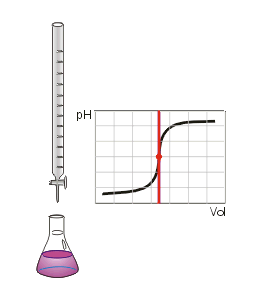
Animation courtesy of By Luigi Chiesa - Draw by Luigi Chiesa, Public Domain, https://commons.wikimedia.org/w/index.php?curid=665615
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning