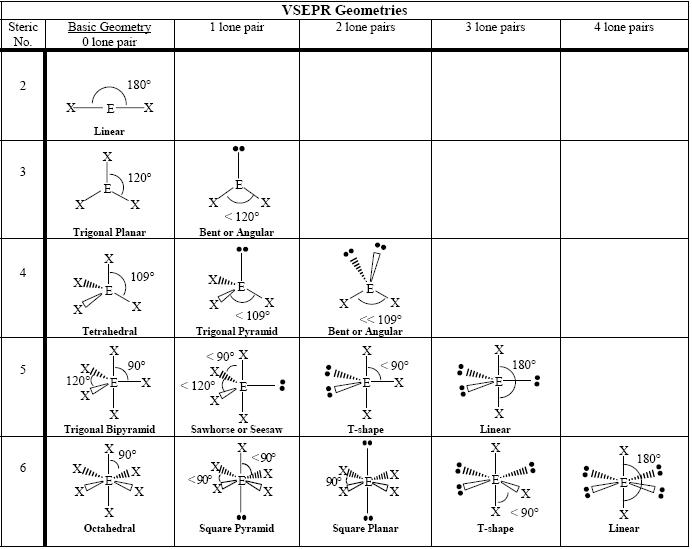
Predicting molecular shape
•Valence Shell Electron Pair Repulsion Theory (VSEPR theory): the electron pairs (lone pairs) in the molecule repel each other and take up positions as far from one another as possible in a 3D space.
•Only bonding pairs of electrons are considered when naming the shape of the molecule.
·Main molecular shape names are: linear, v shaped or bent, pyramidal, tetrahedral and planar.
Diagram- wikimedia commons
•Valence Shell Electron Pair Repulsion Theory (VSEPR theory): the electron pairs (lone pairs) in the molecule repel each other and take up positions as far from one another as possible in a 3D space.
•Only bonding pairs of electrons are considered when naming the shape of the molecule.
·Main molecular shape names are: linear, v shaped or bent, pyramidal, tetrahedral and planar.
Diagram- wikimedia commons