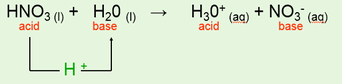ACIDS
•Usually taste sour.
•Are corrosive.
•Are molecular in structure and will dissolve in water.
•Turn litmus from blue to red.
•Are neutralised by bases.
•Examples: HCl, HNO3, H2SO4, CH3COOH.
BASES
•Usually taste bitter.
•Feel slippery.
•Are generally ionic substances.
•Turn litmus from red to blue.
•Are oxides or hydroxides of metals.
•Examples: NH3, NaOH, NH4OH
LOWREY- BRONSTED THEORY
•An acid is a proton donor.
•A base is a proton acceptor.
•A proton is a hydrogen ion H+.
•An acid base reaction involves a proton transfer from the acid to the base.
•Eg: Nitric Acid reacting with water.
•Usually taste sour.
•Are corrosive.
•Are molecular in structure and will dissolve in water.
•Turn litmus from blue to red.
•Are neutralised by bases.
•Examples: HCl, HNO3, H2SO4, CH3COOH.
BASES
•Usually taste bitter.
•Feel slippery.
•Are generally ionic substances.
•Turn litmus from red to blue.
•Are oxides or hydroxides of metals.
•Examples: NH3, NaOH, NH4OH
LOWREY- BRONSTED THEORY
•An acid is a proton donor.
•A base is a proton acceptor.
•A proton is a hydrogen ion H+.
•An acid base reaction involves a proton transfer from the acid to the base.
•Eg: Nitric Acid reacting with water.
IONISATION OF ACIDS
•Ionisation is a reaction in which a molecule reacts with water to produce two or more ions.
•Eg: HCl (l) + H20 (l) → H30+ (aq) + Cl- (aq)
acid base acid base
H30+ is called the hydronium ion.
DISSOCIATION OF BASES
•When ionic bases dissolve in water, they dissociate or separate into their constituent ions.
•They do not actually react with the water.
H20
•Eg: NaOH (s) → Na+ (aq) + OH– (aq)
NEUTRALISATION
•Acid + Base → Salt + Water.
•Eg: hydrochloric acid + sodium hydroxide gives sodium chloride + water.
HCl (aq) +NaOH (aq) → NaCl (aq) + H20 (l)
acid base salt water
•The ionic equation for the above reaction is:
H+(aq)+Cl-(aq)+Na+(aq)+OH-(aq) → Na+(aq)+Cl-(aq)+H20(l)
•Na+ & Cl- are spectator ions and do not take part in the reaction, so ionic equation can be rewritten as:
H+(aq) + OH-(aq) → H20(l)
CONJUGATE ACID AND BASE PAIRS
•When an acid and a base react a conjugate acid and base are formed.
Eg: NH3 (g) + H20 (l) → NH4+ (aq) + OH- (aq)
base1 acid2 acid1 base2
•The conjugate pairs are written as acid / base.
•In the above reaction the conjugate pairs are:
[ NH4+ / NH3 ] and [ H20 / OH- ]
•Conjugate acid-base pairs differ by a proton, H+
•Ionisation is a reaction in which a molecule reacts with water to produce two or more ions.
•Eg: HCl (l) + H20 (l) → H30+ (aq) + Cl- (aq)
acid base acid base
H30+ is called the hydronium ion.
DISSOCIATION OF BASES
•When ionic bases dissolve in water, they dissociate or separate into their constituent ions.
•They do not actually react with the water.
H20
•Eg: NaOH (s) → Na+ (aq) + OH– (aq)
NEUTRALISATION
•Acid + Base → Salt + Water.
•Eg: hydrochloric acid + sodium hydroxide gives sodium chloride + water.
HCl (aq) +NaOH (aq) → NaCl (aq) + H20 (l)
acid base salt water
•The ionic equation for the above reaction is:
H+(aq)+Cl-(aq)+Na+(aq)+OH-(aq) → Na+(aq)+Cl-(aq)+H20(l)
•Na+ & Cl- are spectator ions and do not take part in the reaction, so ionic equation can be rewritten as:
H+(aq) + OH-(aq) → H20(l)
CONJUGATE ACID AND BASE PAIRS
•When an acid and a base react a conjugate acid and base are formed.
Eg: NH3 (g) + H20 (l) → NH4+ (aq) + OH- (aq)
base1 acid2 acid1 base2
•The conjugate pairs are written as acid / base.
•In the above reaction the conjugate pairs are:
[ NH4+ / NH3 ] and [ H20 / OH- ]
•Conjugate acid-base pairs differ by a proton, H+
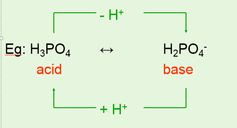
Amphiprotic Acids
•Some substances are capable of acting as acids or bases.
•This means that they are able to donate or accept protons.
•Eg: H20, HSO4-, HCO3- and HS-
HCO3- (aq) + H20 (l) → H30+ (aq) + CO32- (aq)
acid
HCO3- (aq) + NH4+ (aq) → H2CO3 (aq) + NH3 (aq)
base
Polyprotic Acids
•A polyprotic acid can donate more than one proton per molecule of acid.
•Sulfuric acid, H2SO4 is called a diprotic acid as it can donate 2 protons.
•This occurs in 2 stages as shown in the following equations:
H2SO4 (l) + H20 (l) → H30+ (aq) + HSO4- (aq) stage 1
HSO4- (aq) + H20 (l) → H30+ (aq) + SO42- (aq) stage 2
•Monoprotic acid (1 proton) eg: HCl
•Triprotic acid (3 protons) eg: H3PO4
•Some substances are capable of acting as acids or bases.
•This means that they are able to donate or accept protons.
•Eg: H20, HSO4-, HCO3- and HS-
HCO3- (aq) + H20 (l) → H30+ (aq) + CO32- (aq)
acid
HCO3- (aq) + NH4+ (aq) → H2CO3 (aq) + NH3 (aq)
base
Polyprotic Acids
•A polyprotic acid can donate more than one proton per molecule of acid.
•Sulfuric acid, H2SO4 is called a diprotic acid as it can donate 2 protons.
•This occurs in 2 stages as shown in the following equations:
H2SO4 (l) + H20 (l) → H30+ (aq) + HSO4- (aq) stage 1
HSO4- (aq) + H20 (l) → H30+ (aq) + SO42- (aq) stage 2
•Monoprotic acid (1 proton) eg: HCl
•Triprotic acid (3 protons) eg: H3PO4