STUDY CARD AVAILABLE- SEE CONTACT.
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
|
GALVANIC CELLS
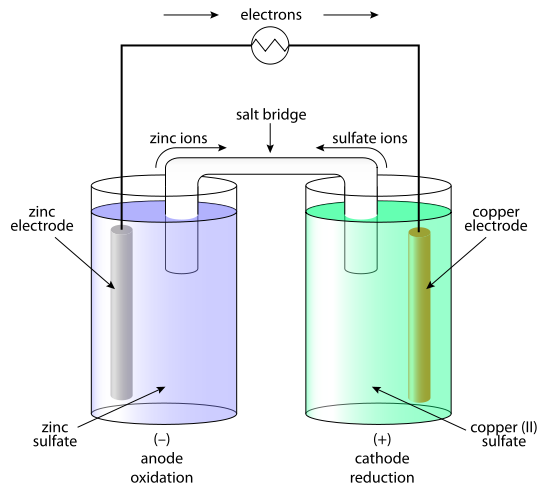
A galvanic cell consists of half reactions that occur in two separate compartments of the cell. The oxidant and the reactant do not come in contact with each other. All electrons must be transferred through the external circuit. This flow creates an electrical current. Chemical energy of the reactants is transferred into electrical energy. HALF CELLS •The species in each half cell form a conjugate redox pair. •In general, if one member of the conjugate pair in a half cell is a metal, it is usually used as the electrode. •If no metal is present, than an inert electrode such as platinum is used. •If one of the conjugate pairs is a gas, a special gas electrode is used. •Half cells also usually contain spectator ions and the particles that make up the solvent. •The electrode where oxidation occurs is called an anode. In galvanic cells the anode, where electrons are produced, is the negative terminal. •The electrode at which reduction occurs is called the cathode. In galvanic cells the cathode, where electrons are consumed, is the positive terminal. SALT BRIDGE •If there was no salt bridge, charge would accumulate in each of the cells. •These accumulations would prevent further reaction and prevent the production of electricity. •The salt bridge contains ions that are free to move so they can balance the charges formed. •Cations move to the cathode and anions move to the anode. •This is called the internal circuit. |
Galvanic cell diagram courtesy of By Hazmat2 - Own work, CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=17902682
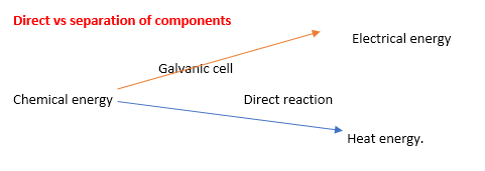
HALF REACTIONS Half Reactions: In each half cell either oxidation or reduction occurs. Oxidation occurs at the anode (-ve) Reduction occurs at the cathode (+ve) Energy transformations in a galvanic cell compared to direct contact. •In a galvanic cell the reactants are separated and do not come into contact. Here the chemical energy is converted into electrical energy. •When reactants are in direct contact, the spontaneous redox reaction (exothermic) converts the chemical energy into thermal (heat) energy. |