|
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
|
|
What is electrolysis?
|
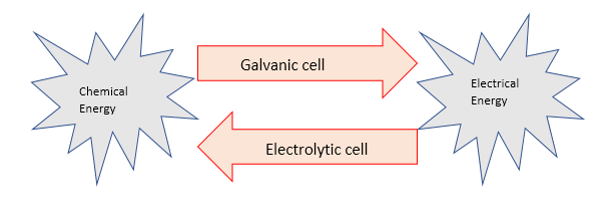
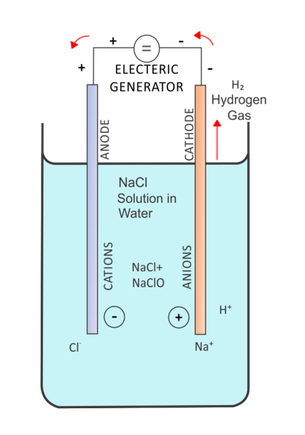
•In electrolysis, a passage of electrical energy from a power source through a conducting liquid causes redox reactions to occur.
•Electrolysis converts electrical energy into chemical energy. They are essentially opposite to the reactions occurring in galvanic cells and would not naturally occur without the application of electrical energy. These reactions are therefore called non-spontaneous. Competition at electrodes•Knowing that oxidation occurs at the anode and reduction occurs at the cathode it is possible to predict the electrode reactions in a particular electrolytic cell.
•Usually there are several chemicals present at each electrode and you must decide which of the possible reactions have the greatest tendency to occur. •In an solution you need to consider that water will also react. During electrolysis•The highest reaction in the electrochemical series that can occur in the forward direction is likely to occur at the cathode.
•That is the strongest oxidant will usually react at the cathode. •The lowest reaction in the electrochemical series that can occur in reverse is likely to occur at the anode. •That is the strongest reductant will usually react at the anode. •It is the opposite to the Galvanic Cell. Steps to follow: 1.Write out all possible reduction reactions that could occur-place these in order of the electrochemical series (include equations involving water for a solution). 2.Circle the oxidants (LHS) and reductants (RHS) present in each equation. 3.Choose the strongest oxidant (most positive E value)- this will undergo reduction- will occur at the negative cathode. 4.Choose the strongest reductant ( bottom right hand corner)- this will undergo oxidation at the positive anode. Generally- the oxidant and the reductant species closest to one another on the series will be the ones that react. |
Electrolytic cell diagram courtesy of Mahdieh.keshmiri (Own work) [CC BY-SA 4.0 (http://creativecommons.org/licenses/by-sa/4.0)], via Wikimedia Commons
|