|
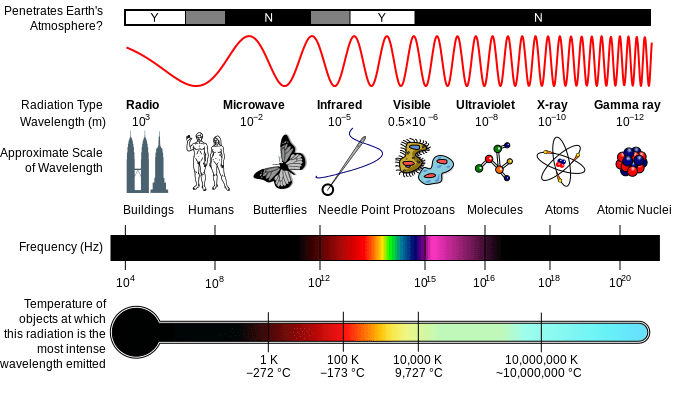
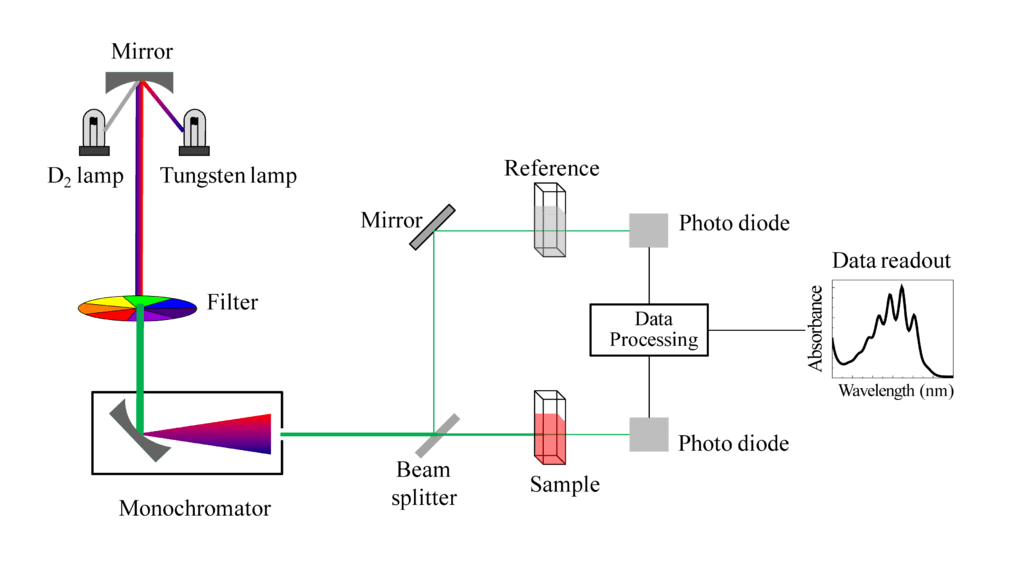
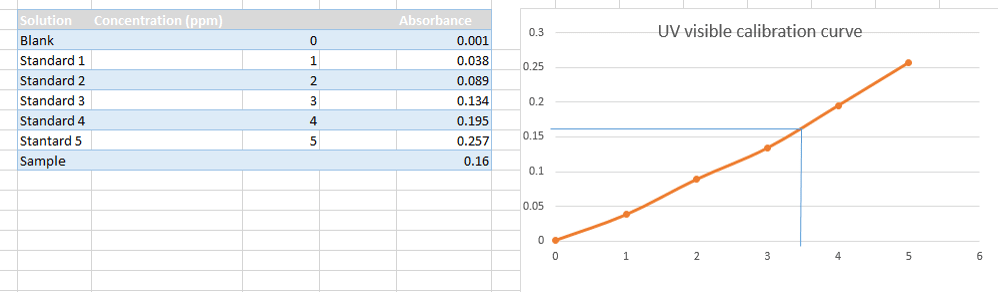
SPECTROSCOPY TECHNIQUES •Spectroscopy techniques can tell us: •The type of atom or molecule present •How much of an atom or molecule is present •The structure and bonding of a molecule. Spectroscopy techniques utilise the fact that: -atoms/molecules absorb and emit electromagnetic radiation of certain energies. -Atoms/molecules undergo change when they absorb electromagnetic radiation. Different parts of the electromagnetic spectrum affect different parts of the atom/molecule. In spectroscopic techniques, the atom or molecule absorbs a certain quantum of energy which causes the atom or molecule to move to a higher energy level. Atoms- movement of electrons to higher level. Molecules- movement of electrons to higher level, movement of molecules to a higher vibrational, rotational and nuclear spin energy levels. The electromagnetic spectrum. •All forms of spectroscopy use part of the electromagnetic spectrum .•All forms of spectroscopy use part of the electromagnetic spectrum. Ultraviolet light is very short wave radiation with high energy, while radio waves have long wavelengths and low energy. COLORIMETRY •Colorimetry or colourimetry is used to determine the concentration of coloured compounds in solution. The absorption is compared to a Calibration Graph- a graph of the different absorptions of standard solutions with known concentrations – to determine the concentration of the solution. UV-VISIBLE SPECTROSCOPY •This method is similar to colorimetry in that it uses a light beam to measure light absorption. • UV – Visible is more effective in that it uses a monochromator to select specific wavelengths to best detect the presence of specific ions. •A light detector identifies which wavelength the sample absorbs and this is compared with the wavelength absorbed by reference samples. •UVVS is particularly useful for detecting metal ions in uncoloured solutions. •Some uses include detecting substances in blood and urine, and determining the amount of coloured dye in plastics. Although UV-Spec is mainly used for finding the concentration of certain elements within a solution, it can also be helpful in qualitative analysis. •When a substance absorbs light it appears coloured. •The colour observed is the complement of the absorbed colour because this is what remains to reach our eyes. |
Animations showing vibration, rotation and translational stretching.
Animations courtesy of http://bestanimations.com/Science/Chemistry/Chemistry2.html Electromagnetic spectrum diagram courtesy of By Inductiveload, NASA - self-made, information by NASABased off of File:EM_Spectrum3-new.jpg by NASAThe butterfly icon is from the P icon set, P biology.svgThe humans are from the Pioneer plaque, Human.svgThe buildings are the Petronas towers and the Empire State Buildings, both from Skyscrapercompare.svg, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=2974242
UV-visible instrumentation diagram courtesy of By Sobarwiki - Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=28009251
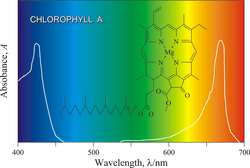
Chlorophyll absorbs strongly at 420nm (blue - complement colour yellow) and 660nm (Red- complement colour blue-green). Looking at the complement colours - the observed colours- it is easy to see why chlorophyll is green in colour..
UV-Visible specta graph courtesy of E. Generalic, http://glossary.periodni.com/glossary.php?en=chlorophyll |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning