Properties of non-metallic substances
•Non-metallic substances only contain non-metal atoms.
•Non-metallic substances usually have low boiling points- since bonding between the compounds is weak.
•Non-metallic substances do not conduct electricity- since they do not contain ions or delocalised electrons.
Covalent bonding
•A covalent bond results when non metallic atoms combine to form either molecules or covalent lattices.
•Covalent bonds involve the sharing of electrons.
Covalent molecules
•Molecules are made up of groups of atoms.
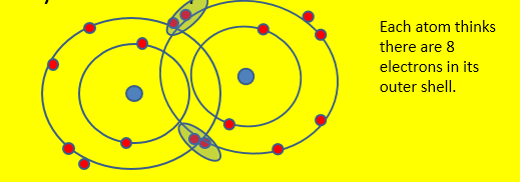
•Adjacent atoms within a molecule share electrons in order to achieve a full outer shell. This happens when an atom is surrounded 8 electrons. This called the octet rule.
•The overall charge on each molecule is zero.
•Examples:
–Water, H20
–Carbon Dioxide, CO2
-Oxygen, O2
Eg. oxygen gas- each atom needs 2 electrons, so they share two pairs.
Lewis Diagrams
·To simplify drawings of atoms, Lewis or electron dot diagrams may be used to represent the outer shell (valence) electrons.
•The outer shell electrons are represented by dots or crosses around element symbol.
•If there are more than 4 valence electrons the dots or crosses are paired up.
•Non bonding electrons or lone pairs are when there are a pair of dots or crosses.
•Bonding electrons are when there are unpaired or single dots or crosses.
(Image wikimedia commons)
•Non-metallic substances only contain non-metal atoms.
•Non-metallic substances usually have low boiling points- since bonding between the compounds is weak.
•Non-metallic substances do not conduct electricity- since they do not contain ions or delocalised electrons.
Covalent bonding
•A covalent bond results when non metallic atoms combine to form either molecules or covalent lattices.
•Covalent bonds involve the sharing of electrons.
Covalent molecules
•Molecules are made up of groups of atoms.
•Adjacent atoms within a molecule share electrons in order to achieve a full outer shell. This happens when an atom is surrounded 8 electrons. This called the octet rule.
•The overall charge on each molecule is zero.
•Examples:
–Water, H20
–Carbon Dioxide, CO2
-Oxygen, O2
Eg. oxygen gas- each atom needs 2 electrons, so they share two pairs.
Lewis Diagrams
·To simplify drawings of atoms, Lewis or electron dot diagrams may be used to represent the outer shell (valence) electrons.
•The outer shell electrons are represented by dots or crosses around element symbol.
•If there are more than 4 valence electrons the dots or crosses are paired up.
•Non bonding electrons or lone pairs are when there are a pair of dots or crosses.
•Bonding electrons are when there are unpaired or single dots or crosses.
(Image wikimedia commons)
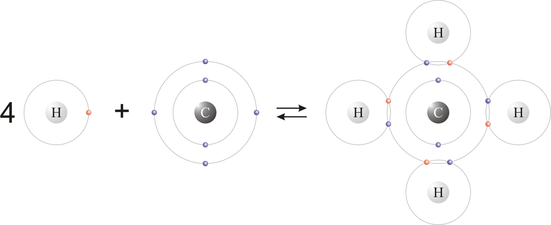
Lewis diagrams for molecules:
•Rules for drawing Lewis Diagrams for molecules:
1. Draw the Lewis Diagrams for each of the atoms in the molecule.
2. Unpaired electrons (bonding electrons) are available for sharing to form a covalent bond.
Paired electrons (lone pairs) do not participate in the bonding.
4. The atom with the most bonding electrons is called the central atom and other atom are bonded to it.
5. In the resulting Lewis diagram, all electrons must be paired.
Diagram courtesy of E. Generalić, http://glossary.periodni.com/glosar.php?hr=kovalentna+veza
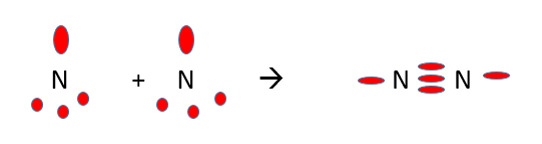
Charge Cloud Representation
•To help visualise the space occupied by bonding and non-bonding pairs of electrons, charge cloud diagrams are commonly drawn.
Example
Nitrogen has 5 electrons in its outer shell. This means there are 3 unpaired electrons and one pair of electrons. Nitrogen is able to bond with itself forming 3 covalent bond and one lone pair.
Eg.
•Rules for drawing Lewis Diagrams for molecules:
1. Draw the Lewis Diagrams for each of the atoms in the molecule.
2. Unpaired electrons (bonding electrons) are available for sharing to form a covalent bond.
Paired electrons (lone pairs) do not participate in the bonding.
4. The atom with the most bonding electrons is called the central atom and other atom are bonded to it.
5. In the resulting Lewis diagram, all electrons must be paired.
Diagram courtesy of E. Generalić, http://glossary.periodni.com/glosar.php?hr=kovalentna+veza
Charge Cloud Representation
•To help visualise the space occupied by bonding and non-bonding pairs of electrons, charge cloud diagrams are commonly drawn.
Example
Nitrogen has 5 electrons in its outer shell. This means there are 3 unpaired electrons and one pair of electrons. Nitrogen is able to bond with itself forming 3 covalent bond and one lone pair.
Eg.