|
. Column Chromatography
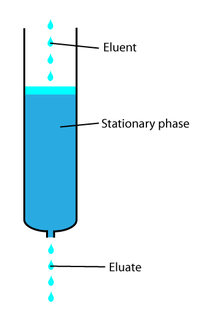
•Sample: The sample is applied to the top of the column and slowly makes its way through, separating as it goes according to how much it is attracted to the stationary and mobile phases. •The time taken for a sample to pass through the column is called the Retention time (Rt). This is similar to an Rf value however it is a record of how long the sample stayed (was retained) in the column. High Performance Liquid Chromatography High performance liquid chromatography is basically a highly improved form of column chromatography. Instead of a solvent being allowed to drip through a column under gravity, it is forced through under high pressures of up to 400 atmospheres. That makes it much faster. HPLC has a solid stationary phase (HPLC tube) which has a much smaller particle size for the column packing material than in standard column chromatography, which gives a much greater surface area for interactions between the stationary phase and the molecules flowing past it. This allows a much better separation of the components of the mixture. The other major improvement over column chromatography concerns the detection methods which can be used. These methods are highly automated and extremely sensitive. HPLC has a digital recorder attached and the chromatogram produced has a peak for each component detected. The time taken for each peak to emerge determines what the component is and the area under the peak can be compared to a standard sample of known concentration to find the unknown concentration Process and Sensitivity: HPLC is very sensitive due to the very small size of the stationary particles in it's column, giving excellent separation. Operates under pressure to assist movement through the column. Uses: HPLC can be both qualitative and quantitative. It is useful for separating and identifying compounds with high molar masses that are not easily vaporised, such as proteins and organic compounds likely to decompose in GC. |
Column chromatography diagram courtesy of By Bakerccm - Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=6827488
Diagram courtesy of By Superchilum - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=8875195
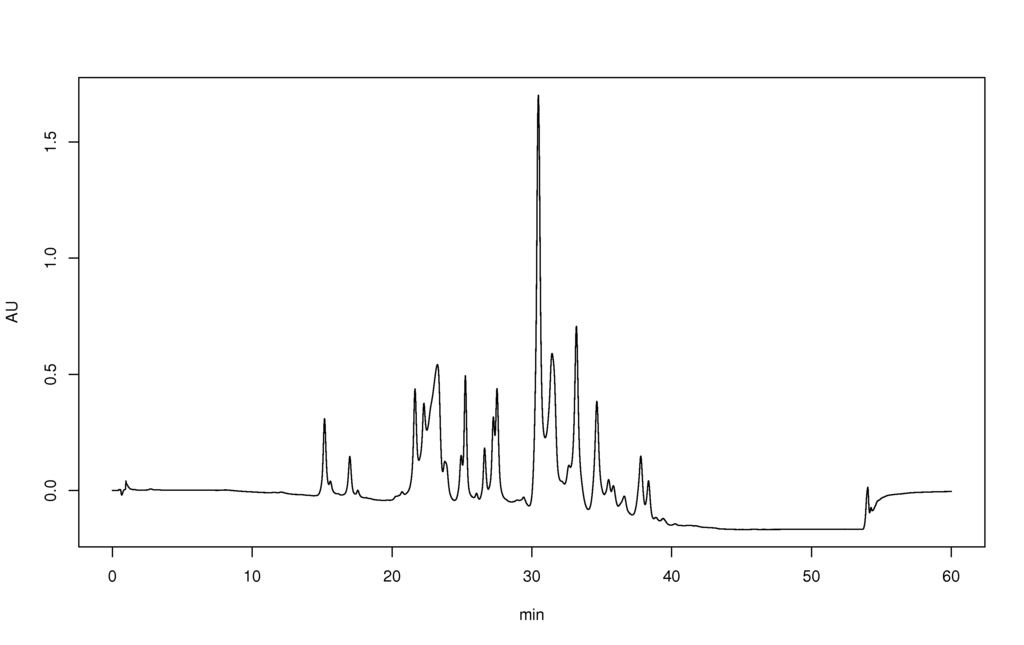
HPLC chromatogram of J'Adore perfume water, as example of complex mixture analysis. Separation on C18 column using almost linear 5 - 100% acetonitrile-water gradient.
Diagram courtesy of By Lukke - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=4867478 |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning