STUDY CARD AVAILABLE- SEE CONTACT.
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646
Key knowledge:
• the common design features of fuel cells including use of porous electrodes for gaseous reactants to increase cell efficiency (details of specific cells not required)
• the comparison of the use of fuel cells and combustion of fuels to supply energy with reference to their energy efficiencies (qualitative), safety, fuel supply (including the storage of hydrogen), production of greenhouse gases and applications
• the comparison of fuel cells and galvanic cells with reference to their definitions, functions, design features, energy transformations, energy efficiencies (qualitative) and applications
• the common design features of fuel cells including use of porous electrodes for gaseous reactants to increase cell efficiency (details of specific cells not required)
• the comparison of the use of fuel cells and combustion of fuels to supply energy with reference to their energy efficiencies (qualitative), safety, fuel supply (including the storage of hydrogen), production of greenhouse gases and applications
• the comparison of fuel cells and galvanic cells with reference to their definitions, functions, design features, energy transformations, energy efficiencies (qualitative) and applications
|
FUEL CELLS
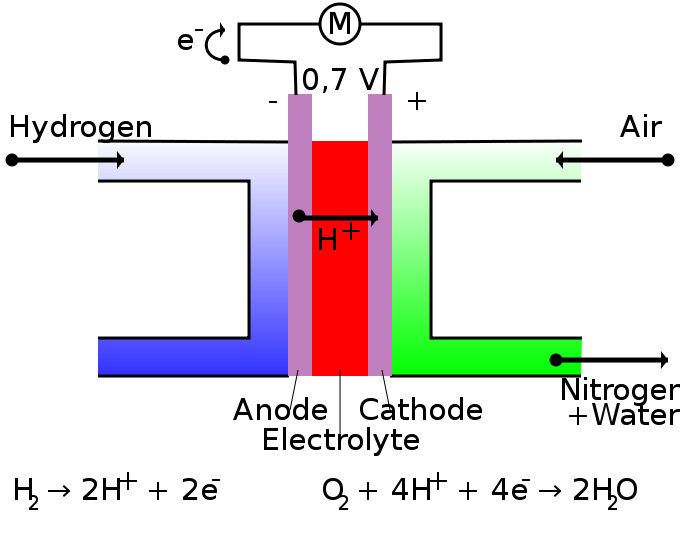
•Cells can be constructed in which the reactants are supplied continuously allowing constant production of electrical energy (Reactants are not stored in the cell) •These devices are called fuel cells. •Fuel cells transform chemical energy directly into electrical energy. •At the anode: H2(g) + 2OH-(aq) --> 2H2O(l) + 2e- •At the cathode: O2(g) + 2H2O(l) + 4e- ---->4OH-(aq) •The overall reaction: 2H2(g) + O2(g) ----> 2H2O(l) •Each cell produces about one volt. •Have porous electrodes that allow reactants to diffuse through them to react with ions in the electrolyte. •They often have catalysts to speed up the reaction. •Higher voltages are obtained by connecting a number of fuel cells in series. •The only by-products are water and heat. •The use of fuel cells in transportation improves fuel efficiency and reduces greenhouse gas and other emissions. ADVANTAGES OF FUEL CELLS •Convert chemical energy directly to electrical energy, better efficiency. •Will generate electricity for as long as fuel is supplied. •Water is a by-product, no greenhouse gases released. •Electricity can be generated on site. DISADVANTAGES OF FUEL CELLS •They are expensive. •Require a constant fuel supply. •Some may use expensive electrolytes and catalysts. •Fuel cells generate a direct current (DC) not AC current used in homes. •Use of fuel cells in transport is limited by lack of facilities for hydrogen storage and distribution. |
Fuel cell diagram courtesy of By HandigeHarry - Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=3923706
HYDROGEN AS A FUEL Steam reforming The majority of hydrogen production is produced from fossil fuels by steam reforming. Steam reforming involves fossil fuels reacting with steam at high temperatures producing carbon monoxide and hydrogen gas. Reaction: Ni CH4(g) + H2O (g) ---> CO(g) + 3H2(g) Reacting the carbon monoxide further gives: Cu CO(g) + H2O (g) ---> CO2(g) + H2(g) Other methods of making hydrogen gas: •Using biogas (Methane) – then use steam reforming to produce hydrogen gas. •Convert water into hydrogen using electricity. 2H2O (l) ---> 2H2(g) + O2(g) Storage Hydrogen is currently stored either by liquid hydrogen or compressed hydrogen. In the future if hydrogen fuel cells are used more extensively storage, delivery and transport of hydrogen will need to be addressed. Hydrogen burns readily and is more dangerous to store than fossil fuels. |

Image courtesy of mediawiki commons- Department of Energy's Energy Efficiency and Renewable Energy Fuel Cell Technologies Program, http://www1.eere.energy.gov/hydrogenandfuelcells/fuelcells/pdfs/fc_comparison_chart.pdf