INSIDE THE ATOM
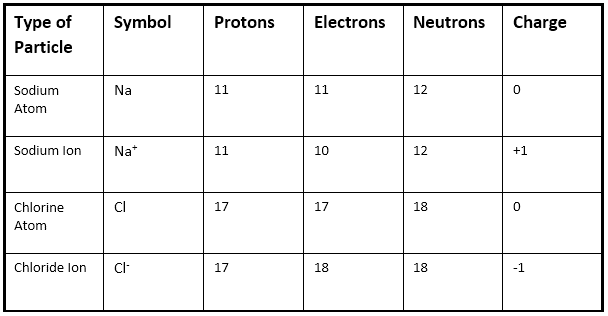
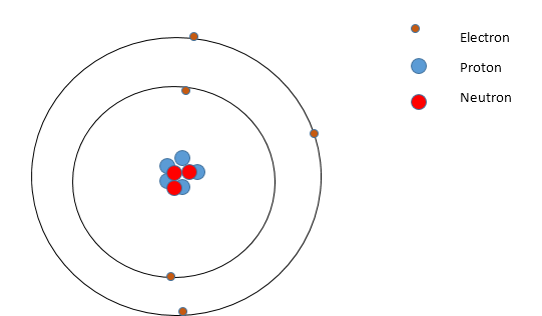
An atom is made up of three subatomic particles
-The electron which carries a negative charge and is found in shells around the nucleus.
-The proton which carries a positive charge and is found in the nucleus.
-The neutron which does not have a charge and is found in the nucleus.
-If an electron is removed or gained from an atom an ion is formed. This is called ionization.
-Nucleus contains protons and neutrons- thus almost all of mass of an atom is in the nucleus.
-Shells where electrons orbit take up most of the volume of an atom.
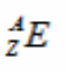
-Atomic Number (symbol Z) of an element is defined as the number of protons in the nucleus of an atom of that element.
-Mass Number (symbol A) is defined as the total number of protons and neutrons in an atom of an element. This is found by subtracting
the atomic number from the mass number (A-Z).
-Isotopes of an element are atoms that have the same atomic number (number of protons) but a different mass number (number of neutrons).
-Mass Number (symbol A) is defined as the total number of protons and neutrons in an atom of an element. This is found by subtracting
the atomic number from the mass number (A-Z).
-Isotopes of an element are atoms that have the same atomic number (number of protons) but a different mass number (number of neutrons).
|
The charge on an atom is the equal to the Protons minus the Electrons.
|
Click to set custom HTML