|
STUDY CARD AVAILABLE- SEE CONTACT.
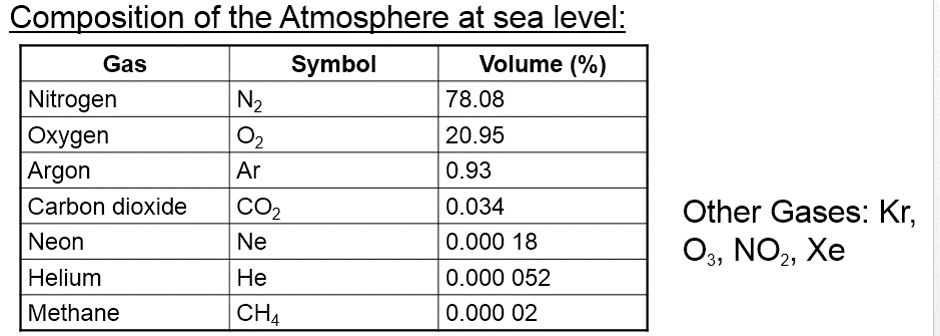
PROPERTIES OF GASES 1.Gases have low densities. 2.Gases fill any container completely and uniformly. 3.Gases are compressible. 4.Gases exert a uniform pressure on all inner surfaces of a container. 5.Gases diffuse (mix) easily and quickly. KINETIC THEORY OF GASES Developed for a fictional ideal gas: 1.Gases are made up of small particles moving constantly and at random in straight lines. 2.Higher temp = more kinetic energy = particles move faster. Kinetic Energy (KE) = 1/2mv^2. m=mass, v=velocity 3.Forces of attraction and repulsion between gas particles are practically zero. 4. Gas particles are very far apart. That is volume of particles is very small compared with volume of gas container. 5. Gas particles collide with each other and walls of their container exerting pressure. Collisions are elastic – energy transferred but total KE is constant (preserved). VOLUME •Volume is the space that a substance occupies. •Units used: 1mL = 1 cm^3 1 L = 1000mL 1 m3 = 1x10^ 6 cm^3 = 1000L PRESSURE Pressure is the force exerted on a unit area by a gas during collision. The more a gas is compressed, the higher the pressure on a container. Our air is made up of a mixture of gases: |
Dalton's partial pressures diagram courtesy of By Max Dodge - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=35993393
|
PARTIAL PRESSURE (DALTON)
•At constant temperature and volume, the total pressure exerted by a mixture of gases is equal to the sum of all the partial pressures of the constituent gases.
Ptotal = P1 + P2 + P3 + …
•At constant temperature and volume, the total pressure exerted by a mixture of gases is equal to the sum of all the partial pressures of the constituent gases.
Ptotal = P1 + P2 + P3 + …
|
•Since pressure is a force it can be written as: Pressure = Force or P = F Area A Units Nm^-2 or 1 Pascal (Pa). UNITS USED FOR GASES •Pressure: (sea level = 1 atmosphere) 760 mmHg = 1 atm = 101 325 Pa 1 bar = 100kPa= 750 mmHg = 0.987 atm Now used at standard conditions –SLC and STP. •Temperature: degrees celsius (oC) kelvin scale (absolute scale) K = oC + 273 or oC = K – 273 •Volume: 1 m^3 = 1000 L = 1 000 000 mL |
|