|
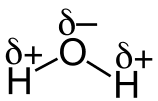
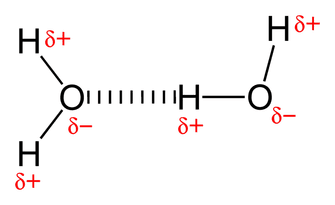
PROPERTIES OF WATER. *Water is a Polar molecule. •Intermolecular forces (forces between molecules) are Hydrogen Bonds. •Hydrogen Bonds are relatively strong form of intermolecular force. •Water is the only substance found commonly in all 3 states (solid, liquid and gas). •Relatively high melting point & boiling point. •Water expands on freezing. •Ice is less dense than water and floats on water. •High heat capacity. Water molecule animation courtesy of Wikimedia - The author of the work and the molecular graphics program used [CC BY-SA 4.0 (http://creativecommons.org/licenses/by-sa/4.0)], via Wikimedia Commons |
Water molecule diagram courtesy of By Jü - Own work, CC0, https://commons.wikimedia.org/w/index.php?curid=15508856
Hydrogen bonding in water- diagram courtesy of Public Domain, https://commons.wikimedia.org/w/index.php?curid=1515822
|

Iceberg picture courtesy of By AWeith - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=51789188