|
•Chromatography can be used for both qualitative and quantitative analysis.
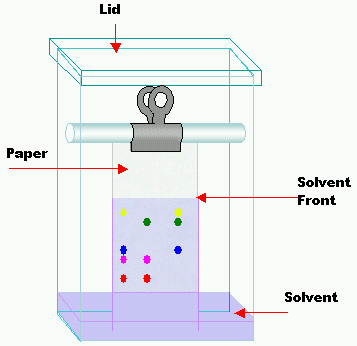
•It can be used to identify numerous inorganic and organic substances such as drugs present in blood and toxic gases in the air. Principles of Chromatography. Chromatography separates the components in a mixture. A sample of the mixture may be dissolved in a solvent. The mobile phase is the liquid or gas that carries the sample forward, and it passes over a solid material, called the stationary phase. Separation occurs as a result of the components' differing abilities to adsorb or desorb (dissolve back into) the mobile phase. The more strongly a component bonds to the stationary phase, the slower the rate of movement of that component over the stationary phase. Chromatography techniques: There are a number of different types of Chromatography: • paper chromatography •thin layer chromatography (TLC) •column chromatography, gas chromatography (GC) •high performance liquid chromatography (HPLC). Paper Chromatography High quality absorbent paper is used as the stationary phase. A solution of the sample is made up and a very small spot is placed onto one end of the paper with a capillary tube. The position of the spot is called the origin. The paper is then placed in a container so that the edge of the paper below the spot is submerged in a solvent. As the solvent (mobile phase) rises up the paper, the components of each sample separate. Thin Layer Chromatography •Similar to paper chromatography except the stationary phase is a fine powder such as alumina spread on a glass or plastic plate. •Paper and TLC are useful for qualitative analysis. Interpreting Chromatograms A chromatogram is the pattern of bands or spots formed on the plate or paper. Individual chemicals can either be determined by: 1. Running known standards on the same chromatogram as the unknown and comparing •In this method it is necessary to have some idea of the chemical present in the sample •The sample and standard must be run on the same chromatogram because the distances moved from the origin will depend on the distance moved by the solvent front. 2. Calculating Rf values of the sample. Rf values The Rf value of components of a mixture is the distance they travel along the stationary phase compared to the distance travelled by the solvent front. •Rf = Distance moved from origin by component Distance moved from origin by solvent •Rf values are always less than one. |
Paper Chromatography diagram courtesy of CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=1418132
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning