Intramolecular and Intermolecular forces
•Intramolecular forces – covalent bonding within molecules.
•Intermolecular forces of attraction only exist in solid and liquid states for that substance.
•Intermolecular forces – forces of attraction between molecules that hold molecules together.
The stronger the intermolecular forces the higher the melting or boiling points
•Three types of intermolecular forces:
1. Dispersion forces ( instantaneous dipole) – weakest (non polar molecules) (Also called London forces or Van der Waals)
2. Dipole – dipole attractions (permanent dipole) – stronger (polar molecules)
3.Hydrogen Bonding (special strong form of dipole – dipole attractions) – strongest
(eg water H20, ammonia NH3, HF)
Dipole-Dipole attractions
•Molecules are polar.
•Permanent dipole.
•Forces of attraction are between the positive end of one molecule and the negative end of another molecule.
•Example: Hydrogen Chloride, HCl
•The closer the dipoles get, the stronger the attraction.
Dispersion forces
•Molecules are non polar.
•Instantaneous dipole.
•Forces of attraction are between the instantaneous positive end of one molecule and the instantaneous negative end of another molecule.
•Example: Hydrogen, H2
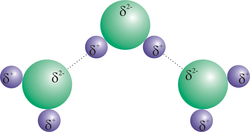
Hydrogen bonding
•Molecules are highly polar.
•Special strong case of dipole – dipole attractions.
•Hydrogen bonding is the force of attraction between the hydrogen atom on one molecule and the fluorine, oxygen or nitrogen atom on another molecule.
•Example: Water, H20
Diagram courtesy of E. Generalić, http://glossary.periodni.com/glosar.php?hr=vodikova+veza
Properties of molecules
•Polar molecules are soluble in polar solvents.
•Example: Ammonia (polar molecule) is soluble in water (polar solvent).
•Non polar molecules are soluble in non polar solvents.
•Example: Methane (non polar molecule) is soluble in carbon tetrachloride ( non polar solvent).
•Molecular substances do not conduct electricity in solid or molten form. (no charged particles).
•Molecular compounds have low melting and boiling points. (molecules held together by dispersion forces or dipole-dipole attractions)
Most molecular substances are soft and easily scratched
•Intramolecular forces – covalent bonding within molecules.
•Intermolecular forces of attraction only exist in solid and liquid states for that substance.
•Intermolecular forces – forces of attraction between molecules that hold molecules together.
The stronger the intermolecular forces the higher the melting or boiling points
•Three types of intermolecular forces:
1. Dispersion forces ( instantaneous dipole) – weakest (non polar molecules) (Also called London forces or Van der Waals)
2. Dipole – dipole attractions (permanent dipole) – stronger (polar molecules)
3.Hydrogen Bonding (special strong form of dipole – dipole attractions) – strongest
(eg water H20, ammonia NH3, HF)
Dipole-Dipole attractions
•Molecules are polar.
•Permanent dipole.
•Forces of attraction are between the positive end of one molecule and the negative end of another molecule.
•Example: Hydrogen Chloride, HCl
•The closer the dipoles get, the stronger the attraction.
Dispersion forces
•Molecules are non polar.
•Instantaneous dipole.
•Forces of attraction are between the instantaneous positive end of one molecule and the instantaneous negative end of another molecule.
•Example: Hydrogen, H2
Hydrogen bonding
•Molecules are highly polar.
•Special strong case of dipole – dipole attractions.
•Hydrogen bonding is the force of attraction between the hydrogen atom on one molecule and the fluorine, oxygen or nitrogen atom on another molecule.
•Example: Water, H20
Diagram courtesy of E. Generalić, http://glossary.periodni.com/glosar.php?hr=vodikova+veza
Properties of molecules
•Polar molecules are soluble in polar solvents.
•Example: Ammonia (polar molecule) is soluble in water (polar solvent).
•Non polar molecules are soluble in non polar solvents.
•Example: Methane (non polar molecule) is soluble in carbon tetrachloride ( non polar solvent).
•Molecular substances do not conduct electricity in solid or molten form. (no charged particles).
•Molecular compounds have low melting and boiling points. (molecules held together by dispersion forces or dipole-dipole attractions)
Most molecular substances are soft and easily scratched
