The Biological Importance of Proteins
•Proteins are found in every cell and are fundamental to cell structure and operation. The protein in food is the only major nutrient that provides us with a source of nitrogen. Nitrogen is needed to make specific proteins and other nitrogen- containing compounds present in the human body.
There are many different proteins which serve different purposes in the human body. Such as enzymes, hormones and those for structual purposes within the body (muscle and bone for example).
There are many different proteins which serve different purposes in the human body. Such as enzymes, hormones and those for structual purposes within the body (muscle and bone for example).
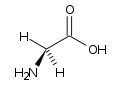
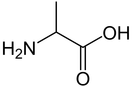
Amino acidsProteins are polymers built up from small monomer molecules called amino acids. Two of the simpler amino acids are glycine and alanine.
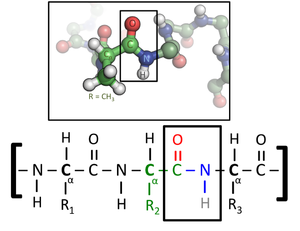
Every amino acid has an amino group (NH2) and a carboxy group (COOH). They have the general formula H2N-CHZ-COOH The major difference between amino acids is the atoms that make up the side chain (Z group). These amino acids are known as 2-amino acids or alpha-amino acids. Amino Acids are soluble in water as they have polar amino and carboxy functional groups. In solution the amino group can act as a base and the carboxy group acts as an acid. In solution: Neutral pH/solution: +H3N-CHZ-COO- zwitterions or dipolar ion •A proton has been lost from the acidic carboxy group and accepted by the basic amino group. Acidic pH/solution: +H3N-CHZ-COO- (aq) + H3O+ (aq) <--> +H3N-CHZ-COOH (aq) + H2O (l) Basic pH/solution: +H3N-CHZ-COO- (aq) + OH- (aq) <--> H2N-CHZ-COO- (aq) + H2O (l) As amino acids have the ability to react with both acids and bases, they can act as buffers. Buffers minimise the effect that the addition of H+ or OH- ions have on acidity- important in biochemical processes. Protein structureWhen a molecule that contains a carboxyl group, COOH, combines with a molecule containing an amine group, NH2, a condensation reaction occurs to form an amide functional group, -CONH-, that links the two molecules. A water molecule is also formed. The resulting linkage is called an amide or peptide linkage.
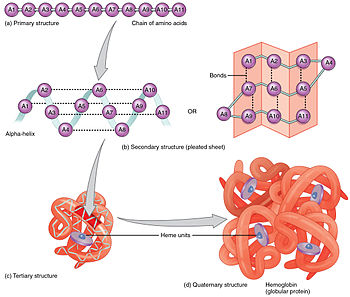
Molecules made from amino acids are often called peptides. Dipeptide- two amino acids joined together. Tripeptide- three amino acids joined together. Polypeptide- polymer made from peptide monomers. Protein- polymers made up of more than 50 amino acids. Proteins differ from one another in the number, type, and sequence of their constituent amino acids. The role that any protein fulfils in an organism depends on its shape. Primary structureThe order that the amino acids that make up the protein are joined in.
Secondary structureThree-dimensional shapes by coiling, folding or pleating of proteins chains. This is caused by the hydrogen bonding between the –CO and N-H groups in neighbouring amino acids.
Tertiary structureThe Z groups in the amino acid units of the protein molecule influence the overall 3D shape. Four types of attractions are important in chain folding:
-Hydrogen bonds between Z groups and –O-H or –N-H bonds. -Ionic interactions between Z groups that contain –NH3+ and other with –COO- -Covalent cross-links eg. Disulfide links (-S-S-), such as insulin. -Dispersion forces between non-polar Z groups. |
Glycine diagram By Original uploader was Paginazero at it.wikipedia - Originally from it.wikipedia; description page is/was here., Public Domain, https://commons.wikimedia.org/w/index.php?curid=2903331
Alanine diagram By Sten André - Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=13579873
Amino acids structures- VCE chemistry data booklet.
Peptide linkage diagram By Chemistry-grad-student - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=16345656
Structures of proteins diagram By OpenStax College - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30131163
|