ORGANIC CHEMISTRY REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-revision-organic-chemistry-12042644
Reaction pathways
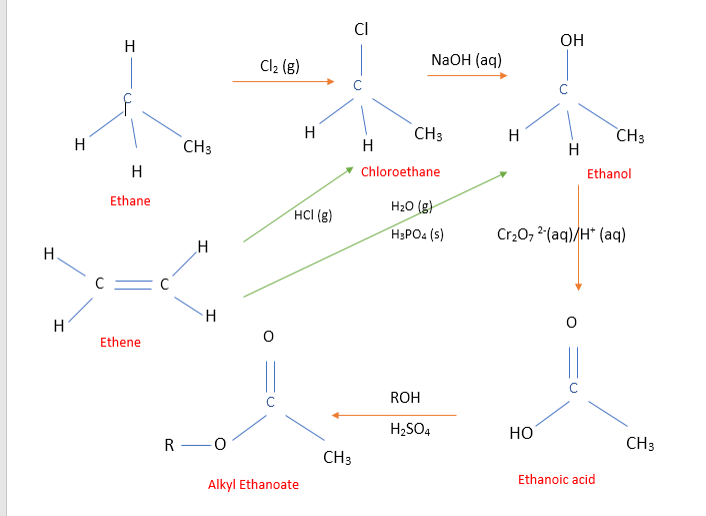
Organic chemists like to create molecular structures that have exactly the right properties needed for a particular purpose. They do this by devising efficient methods for converting a readily available starting material (like an alkane or alkene) into the product they want.
The reaction pathway selected needs to take into account the yield and purity of the products and also minimise any unwanted side-products and waste materials. Time and cost factors also need to be considered.
The reaction pathway selected needs to take into account the yield and purity of the products and also minimise any unwanted side-products and waste materials. Time and cost factors also need to be considered.
|
E.g. How do you form ethyl propanoate (an ester) from ethene and propane?
•Being an ester you need to react a carboxylic acid with an alkanol – make these first! •ethene → ethanol - addition or addition/substitution reactions •propane → propanoic acid - substitution reactions! •Now you have the reactants needed, react them! Esterification reaction Yield•The yield is the amount of product you expect to get. Sometimes, other bi-products form and reduce the yield – not exactly what chemists want!
•In multiple-step processes, a lower yield in one step will affect the later stages. •E.g. A → B yields 80%. B → C yields 70%. Overall A → C yields 80% x 70% X 100% = 56% Atom Economy•Atom economy looks at how may atoms in the reactants end up in the desired product.
•Calculated using: atom economy = molar mass of desired product x100 molar mass of all reactants |
|