ORGANIC CHEMISTRY REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-revision-organic-chemistry-12042644
Stereoisomers
|
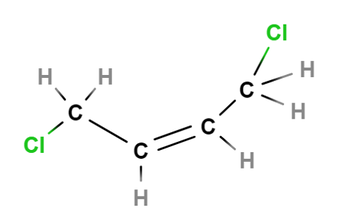
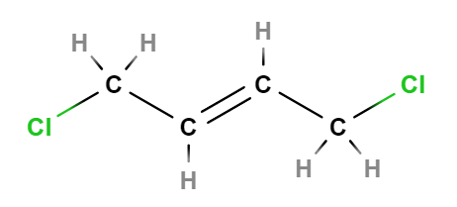
•Stereoisomers- isomers in which the atoms in two molecules are connected in the same order, but have different arrangements in space. Ie. Rotation through bonds. There are 2 types of stereoisomers: 1.Optical isomers 2.Geometric isomers. Optical isomers- stereoisomers in which there is a different placement of groups around one atom in the molecule. Called optical isomers because of the way they interact with plane-polarised light. Chirality Substances that are optical isomers are said to be chiral- mirror images of each other. Eg. Methane- where carbon is the chiral centre. A pair of chiral molecules are called enantiomers. Properties of enantiomers Identical physical properties- except with plane-polarised light. Interact differently with other chiral molecules. Geometric isomers- these isomers occur when there is restricted rotation somewhere in a molecule.eg. Carbon carbon double bond. Types: Cis- groups appear on the same side of the double bond. Trans-groups appear on different sides of the double bond. Images of cis and trans-1,4-Dichloro-2-butene
Images created using Molview |
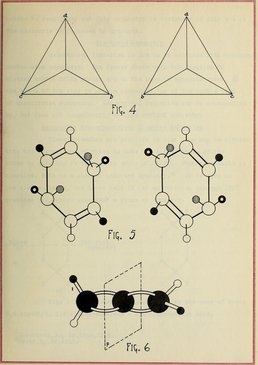
Optical isomerism diagram By Internet Archive Book Images [No restrictions], via Wikimedia Commons
Dinitrogen diflouride cis and trans molecules diagrams By R8R Gtrs (Own work) [CC0], via Wikimedia Commons
|