|
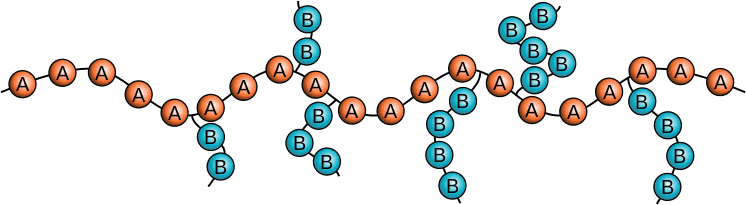
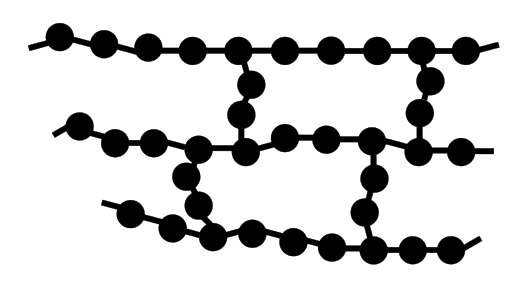
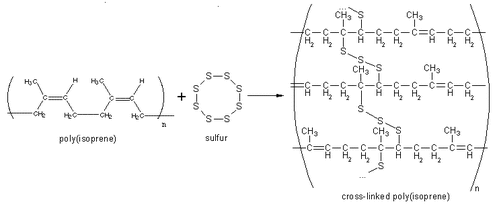
POLYMER PROPERTIES •Polymer properties are determined by the monomers used, the addition of additives, chain length and the extent branching. Polymers can be: •Non-polar •Polar •Be made up from copolymers •Be conductive POLYMERS WITH SIDE BRANCHES •When a hydrogen atom on ethene is replaced by –CH3 group the molecule is propene. •The polymer formed from propene is called polypropene and the –CH3 is called a side branch. •Increased branching in polymers is brought about by the use of higher temperatures and pressure. POLYMERS WITH CROSS-LINKS •Forces between the chains in thermosoftening polymers are mainly weak dispersion forces. •If a small amount of cross linking (bonding) between the chains is introduced an elastomer (eg vulcanised rubber) is produced. •When a large amount of cross linking is introduced, a rigid thermosetting polymer results. RUBBER •Rubber is an addition polymer that occurs naturally. •The monomer is isoprene or 2-methylbuta-1,3-diene. •Properties: 1.Not elastic. 2.Very brittle when cold. 3.Sticky when hot. VULCANISED RUBBER •Natural rubber undergoes an industrial process called vulcanisation to modify its properties. •Natural rubber is mixed with sulfur and heated and the sulfur atoms form cross links between the chains. •Vulcanised rubber is elastic and more resistant to temperature changes. |
Copolymer image courtesy of By Minihaa - Own workThis vector image was created with Inkscape., CC0, https://commons.wikimedia.org/w/index.php?curid=37637259
Elastomer image courtesy of By Koh Wei Teck - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=46692959
Vulcanisation diagram courtesy of By Firey Deity at English Wikipedia - Transferred from en.wikipedia to Commons by Smooth_O using CommonsHelper., Public Domain, https://commons.wikimedia.org/w/index.php?curid=4132922
|
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning