SPECTROSCOPY REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-spectroscopy-revision-poster-ir-nmr-and-ms-12042499
Nuclear magnetic Resonance (NMR)
|
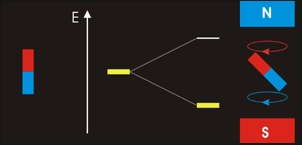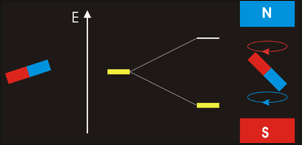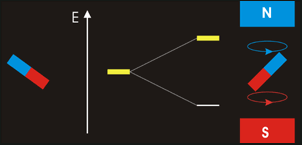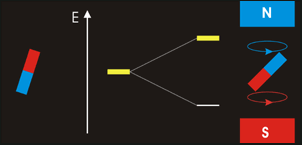
In NMR spectroscopy energy in the form of ‘radio-frequency’ radiation is absorbed by the nuclei in the sample. This causes nucleus of an atom to be excited from its lowest energy spin state to the next higher one.
The time it takes for excited nuclei to relax back (return) to their ground state is called the ‘relaxation’ time. All nucleons (protons and neutrons) are considered to have have two spin states with quantum numbers of +½ (up, ↑)and -½ (down, ↓). Both states are equally likely. In nuclei - with even atomic or mass number the spins cancel out so there is no overall magnetic effect (magnetic moment). In nuclei with an odd total number of nucleons – e.g.1H, 13C – the nucleus has a residual spin and can align itself with or against an external magnetic field. A situation where the nucleus spin is aligned with the magnetic field is lower in energy than the one in which it opposes the magnetic field. The NMR spectrometer•Essentials components of an NMR spectrometer are
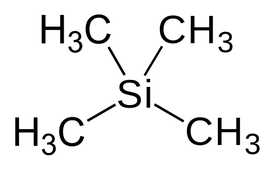
- a powerful magnet, - a radio-frequency generator, and - a radio-frequency detector. - attached computer etc as well, of course, as a way of getting the sample being analysed into the spectrometer • The sample is dissolved in a solvent, most commonly CDCl3 or D2O, and placed in a sample tube which is then suspended in the magnetic field and set spinning. The D in CDCl3 and D2O stands for deuterium – the 2H isotope of hydrogen. Deuterium atoms have sufficiently different magnetic properties from ordinary hydrogen that they don't produce peaks in the area of the spectrum that we are usually looking at. Tetramethylsilane (TMS)The electrons in the C-H bonds are closer to the hydrogens in this compound than in almost any other one. That means that these hydrogen nuclei are the most shielded from the external magnetic field, and so you would have to increase the magnetic field by the greatest amount to bring the hydrogen nuclei back into resonance. Alternatively for a specific applied field the H nuclei in TMS would come into resonance at the lowest frequency.
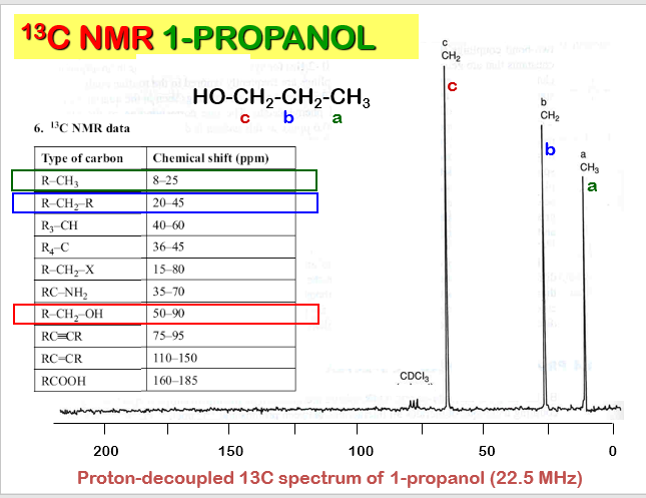
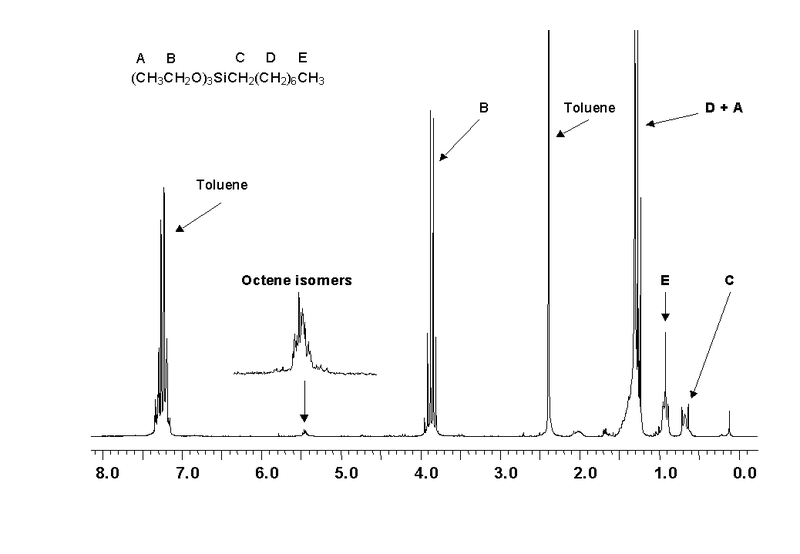
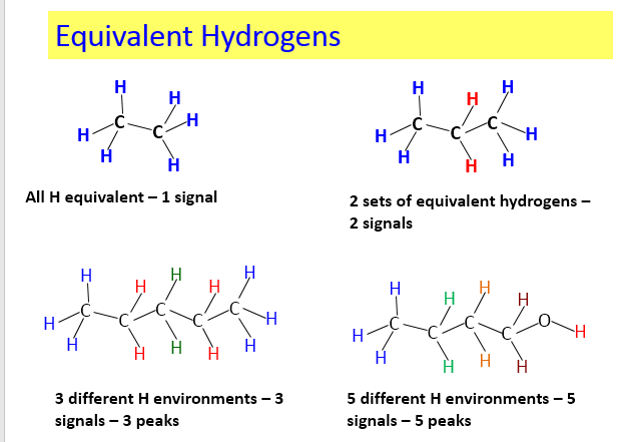
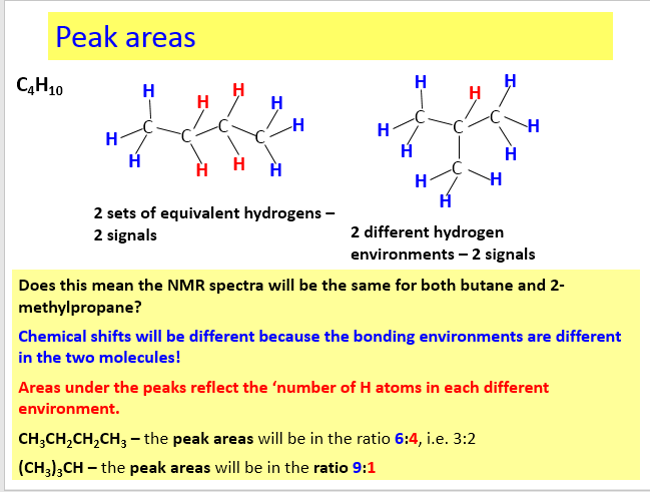
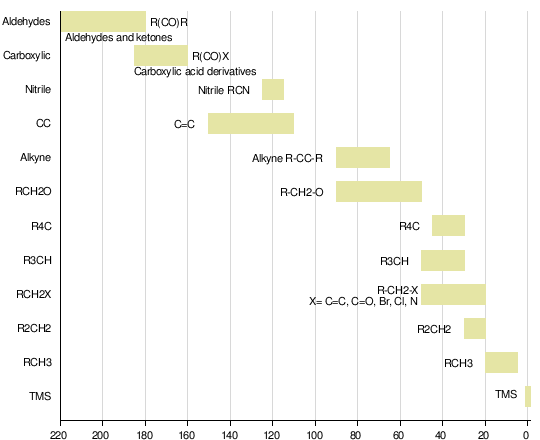
The net effect of this is that TMS produces a peak on the spectrum at the extreme right-hand side. Almost everything else produces peaks to the left of it. The 1H-NMR spectrum of a compound, shows how far the resonance signals of its hydrogen atoms ‘shift’ from the signal generated by the 12 H atoms in tetramethyl silane (TMS). The 13C-NMR spectrum of a compound, shows how far the resonance signals of its carbon atoms ‘shift’ from the signal generated by the 4 C atoms in TMS. Effect of shielding electronsThe most important result of the NMR frequency for applications of NMR is the 'shielding' effect of the surrounding electrons. In general, this electronic shielding reduces the magnetic field at the nucleus (which is what determines the NMR frequency). As a result the energy gap is reduced, and the frequency required to achieve resonance is also reduced. This shift in the NMR frequency is called chemical shift. If a nucleus in a specific chemical group is shielded to a higher degree then its NMR frequency will be shifted "upfield" (that is, a lower chemical shift), whereas if it is less shielded then its NMR frequency will be shifted "downfield" (that is, a higher chemical shift). The closer the signal is to TMS the closer the electrons are to the H atoms associated with the signal. The closer the electrons are to the H atoms the less the effect of the applied magnetic field-so the hydrogen atoms are more shielded. Data for H and C NMR below- source VCE Chemistry data book. See link below. Equivalent hydrogens and peak areasEquivalent hydrogens have the same environment. That is they are joined to the same groups. See the third diagram in the equivalent hydrogen diagrams. The two CH2 groups are the same as both are joined to one CH3 group and one CH group- so they are equivalent.
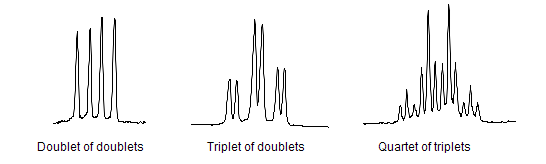
The areas under the peaks tell you how many hydrogens there are in each enviroment. Splitting patternsSplitting patterns follow the n+1 rule.
Here look at the number of H's on the adjacent C - then add 1- this will give you the splitting pattern. Example- Looking at the chemical shift diagram on the right. CH3 group (green) - adjacent carbon has 0 H's- so add 1- so the peak for CH3 is a single peak. CH2 group (red) - adjacent carbon has 3 H's - so add 1 more- 4. This will give a quartet for the CH2 group. CH3 group (blue) - adjacent carbon has 2 H's - so add 1- 3. This CH3 group is a triplet. 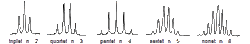
Coupling diagrams By ChemistHans at English Wikipedia - Transferred from en.wikipedia to Commons by T.vanschaik using CommonsHelper., Public Domain, https://commons.wikimedia.org/w/index.php?curid=8476261
Carbon NMRIn carbon NMR - look at each carbon present- see if the environments are the same. Each group will have a chemical shift - depending on what they are bonded two. More carbons in the same enviroment the larger the areas under the peak.
|
NMR spin states animation By Darekk2 (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons
NMR splitting diagram By JBancroftBrown (Own work by the original uploader) [Public domain], via Wikimedia Commons
NMR shift•At the molecular level, local magnetic fields shield hydrogen from the applied magnetic field and so change the frequency of rf radiation needed to bring the nuclei into resonance (where nuclei in a magnetic field absorb and re-emit electromagnetic radiation)
• The effective magnetic field experienced by a 1H nucleus is the applied field less the local magnetic field (due to electrons). The same applies to carbon nuclei in 13C NMR spectroscopy. Tetramethylsilane (TMS) diagram By Jynto [Public domain], via Wikimedia Commons
H MNR spectrum By Przykładowe widmo 1H NMR z mojej własnej publikacji {{GFDL}} from pl wp (usuń) (bież) 14:13, 15 maj 2004 . . Polimerek (Dyskusja) . . 1038x719 (8874 bajtów) (Przykładowe widmo 1H NMR z mojej własnej publikacji)
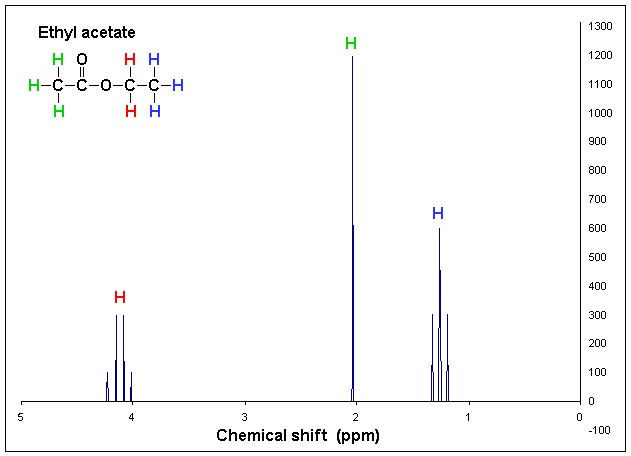
Chemical shift diagram By 1H_NMR_Ethyl_Acetate_Coupling_shown.GIF: T.vanschaik derivative work: H Padleckas [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0) or GFDL (http://www.gnu.org/copyleft/fdl.html)], via Wikimedia Commons
SummaryTo summarise, there are four levels of info in H-NMR:
1.How many types (environments) of H? 2.How many H’s in each environment? 3.What is each type (its chemical shift)? 4.What coupling patterns exist?- n+1 rule Image from Wikipedia
|