ORGANISATION
•Modern periodic table has chemical elements arranged in order of increasing atomic number.
•Elements are arranged in rows and columns in relation to their electronic configuration and also their chemical properties.
•The chemical properties of an element depend on the number of their outer shell electrons.
•Periods are horizontal rows that indicate the number of shells each atom of that element has
•Groups are vertical columns that represent the number of outer shell electrons, valence electrons, in the atoms of the elements in that group.
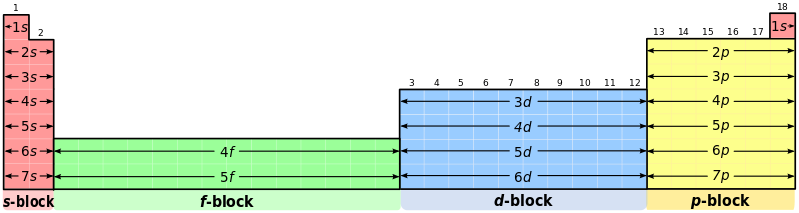
PERIODIC TABLE BLOCKS
There are 4 main blocks in the periodic table:
1.s block = Group I and Group II elements.
Outer shell electronic configuration is s^1 or s^2.
2. p block = Group III to Group VIII elements.
Outer shell electronic configuration is s2p^1 to s^2p^6.
3. d block = Transition Metals.
Outer shell electronic configuration is s^2d^1 to s^2d^10.
4. f block = Lanthanides and Actinides.
Outer shell electronic configuration is f^1 to f^14.
.
•Modern periodic table has chemical elements arranged in order of increasing atomic number.
•Elements are arranged in rows and columns in relation to their electronic configuration and also their chemical properties.
•The chemical properties of an element depend on the number of their outer shell electrons.
•Periods are horizontal rows that indicate the number of shells each atom of that element has
•Groups are vertical columns that represent the number of outer shell electrons, valence electrons, in the atoms of the elements in that group.
PERIODIC TABLE BLOCKS
There are 4 main blocks in the periodic table:
1.s block = Group I and Group II elements.
Outer shell electronic configuration is s^1 or s^2.
2. p block = Group III to Group VIII elements.
Outer shell electronic configuration is s2p^1 to s^2p^6.
3. d block = Transition Metals.
Outer shell electronic configuration is s^2d^1 to s^2d^10.
4. f block = Lanthanides and Actinides.
Outer shell electronic configuration is f^1 to f^14.
.
Image- wikimedia commons