|
RATE AND EQUILIBRIUM REVISION PLACE MAT - https://www.tes.com/teaching-resource/vce-chemistry-unit-3-and-4-rate-and-equilibrium-revision-11987322
WHAT IS EQUILIBRIUM?
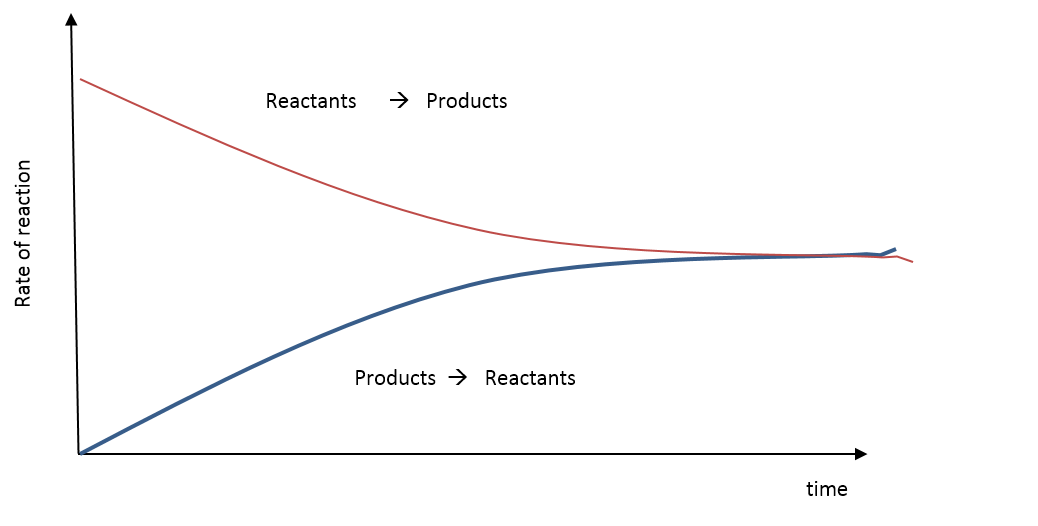
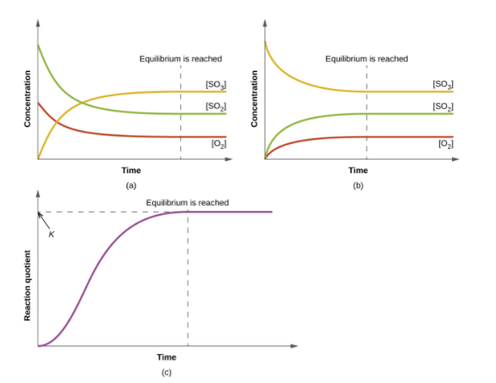
•Chemical Equilibrium occurs when the quantities of reactants and products in a reaction remain unchanged. This means that a reaction may not proceed to completion. The surroundings will have an effect on the equilibrium of a reaction. There are two types of systems: •Open system- a system in which energy and matter can be exchanged with the surroundings. Eg. Fire burning outside. •Closed system- a system in which only energy can be exchanged with the surroundings. Eg. Environment inside a space station. Equilibrium reactions occur in closed systems. Reactions can be irreversable and reversable. For example cooking an egg is an irreverable reaction whereas melting ice and then refreezing it is a reversable reaction. An equilibrium reaction is one in which there is a forward and reverse reaction occuring at the same time. The reaction will reach a balance- be at equilibrium. A double headed arrow is used to show equilibrium of a chemical reaction. DYNAMIC EQUILIBRIUM Dynamic equilibrium is reached when the rate of forward and reverse reactions are equal. At this point all concentrations of reactants/products and the temperature remain constant. Example: Using the Haber Process reaction as an example: N2(g) + 3H2(g) <-----> 2NH3(g) When the reactants are first mixed the forward reaction occurs and ammonia gas is produced. N2(g) + 3H2(g)→ 2NH3(g) As the reactants are used up in the reaction the amount of ammonia produced increases. As the ammonia is being formed, the ammonia molecules react to form nitrogen and hydrogen gases. (The back reaction occurs) 2NH3(g) →N2(g) + 3H2(g) When the forward and reverse reactions occur at the same rate, equilibrium has been reached. What does the Equilibrium Law tell us? The equilibrium constant can tell us the extent of the reaction. •For values of K between 10^-4 and 10^4, there will be significant amounts of reactants and products still present. •For very large values of K > 10^4 , the equilibrium mixture will be mostly products. •For very small values of K < 10^-4 , the equilibrium mixture will be mostly reactants. The effect of temperature. •By experimentation it has been shown that the value of the equilibrium constant for a particular reaction depends only upon temperature. •As the temperature increases: -For exothermic reactions, the amount of the products decreases and so the value of K decreases. -For endothermic reactions, the amount of the products increases and so the value of K increases. |
Click to set custom HTML
Diagram courtesy of OpenStax
THE EQUILIBRIUM LAW Shows a relationship between the concentrations of the reactants and the concentration of the products at equilibrium. The ratio of the products to the reactants found to give the equilibrium constant - K. K = [products] [reactants] For the equation: aA + bB <------> cC + dD K = [C]^c [D]^d [A]^a [B]^b Example: Find the value of the equilibrium constant K, for the reaction below, 2 NO (g) + O2 (g) <-----> 2 NO2 (g) where there is 0.082 M oxygen, 0.050 M nitric oxide and 0.010 M nitrogen dioxide. Solution: Kc = [NO2]2 [NO] 2 x [O2] K= (0.010)2 / [(0.050)2 x (0.082)] = 0.488M^-1 |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning