|
PROPERTIES OF ELEMENTS
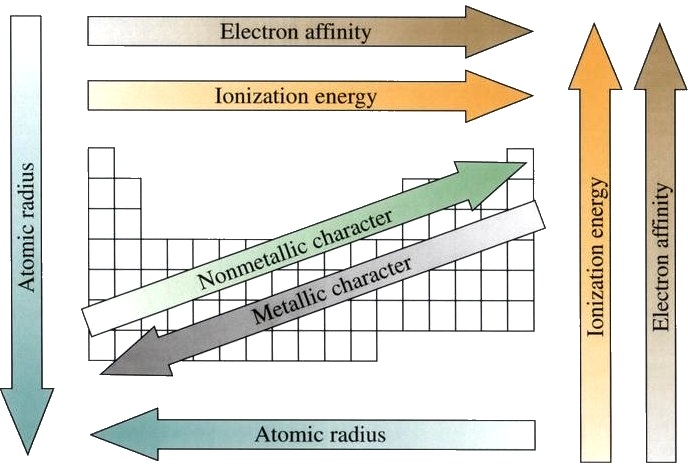
•Atomic Radius: half the distance between 2 nuclei in a molecule. •Ionisation Energy: energy required to remove an electron from a gaseous atom. •Electronegativity: the atoms electron attracting power. •Metallic / Non metallic Character- metals form positive ions (small number of electrons in the outer shell), non-metals form negative ions (large number of electrons in the outer shell). TRENDS WITHIN GROUPS •From top to bottom in a group the number of filled electron shells in atoms increases. •The outer shell electrons are further away from the nucleus and are held less tightly. From top to bottom in a group: •Atomic radius ↑; •First ionisation energy ↓ •Electronegativity ↓ •Metallic properties ↑; Non metallic properties ↓ TRENDS ACROSS PERIODS •Outer shell electrons, because of shielding by inner shell electrons, do not feel the full effect of the nucleus. •The core charge gives an indication of the nuclear pull on outer shell electrons. •Core charge = protons – inner shell electrons In period 3 core charge for: •Na (11p, ec 2,8,1) is +1 (11 – 10) •Cl (17p, ec 2,8,7) is +7 (17 – 10) •Moving from left to right across a period, the core charge experienced by outer shell electrons increases. •The effective nuclear pull is stronger and the electrons are drawn closer to the nucleus and held more tightly. From left to right across a period: •Atomic radius ↓; •First ionisation energy ↑ •Electronegativity ↑ •Metallic properties ↓; Non metallic properties ↑ |
Image - wikimedia commons
|
Image- wikimedia commons