|
STUDY CARD AVAILABLE- SEE CONTACT.
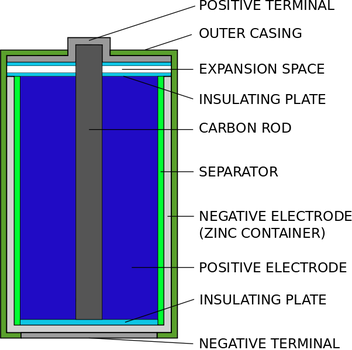
GALVANIC, FUEL CELLS AND ELECTROLYSIS REVISION PLACE MAT- https://www.tes.com/teaching-resource/vce-chemistry-galvanic-and-electrolysis-revision-12042646 PRIMARY CELLS •Batteries such as dry cells, alkaline cells and button cells have one common feature; they are non-rechargeable. •Cells that cannot be recharged are called primary cells. •In primary cells, the products slowly migrate away from the electrodes and are consumed by side reactions occurring in the cells. ZINC -CARBON DRY CELL •This was the first small scale source of electrical energy. •An electrolyte composed of a moist paste of zinc chloride and ammonium chloride and it plays the same role as the salt bridge. •At the anode (-), oxidation of the zinc case produces the electrons. Zn (s) -----> Zn^ 2+ (aq) + 2e- •At the cathode (+), reduction of manganese dioxide. 2MnO2 (s) + 2NH4^+ (aq) + 2e- ---> Mn2O3(s) + 2NH3 (aq) + H2O (l) •A new cell produces about 1.5 volts, but this diminishes during use. •To maintain a net forward reaction, the reaction products must migrate away from the electrodes. •During use, the build up of products at the electrodes slows and can stop the forward reaction. •This is known as polarisation. •If a cell is allowed to rest, some of the products migrate away from the electrodes. •However, once the cell reaches equilibrium, the cell will be flat. ALKALINE CELLS •Have largely replaced Zinc-Carbon Cells. •The chemical reaction within an alkaline cell is similar to the Zinc-Carbon cell but the construction is totally different. •At the anode, zinc powder around the central metal rod is oxidised and once the ion is formed it reacts immediately with the OH- ions in the electrolyte to form zinc hydroxide. Zn (s) -----> Zn^ 2+ (aq) + 2e- Zn (s) + 2OH- (aq) ---> Zn(OH)2 (s) + 2e- •At the cathode, manganese dioxide is reduced. 2MnO2(s) + H2O (l) + 2e- ----> Mn2O3 (s) + 2OH-(aq) •The improvements in this cell give it about five times the life of the Zinc-Carbon cell. RECHARGABLE CELLS AND BATTERIES •Rechargeable cells are known as secondary cells or accumulators. •To recharge a cell, the products of the reaction must be converted back into the original reactants. •This is done by connecting the cell to a charger, a source of electrical energy, which has a potential difference greater than the potential difference of the cell. •Electrical energy supplied is converted into chemical energy in the cell. •In order for it to be possible to regenerate the reactants, the products formed in the cell during discharge must remain in contact with the electrodes in a convertible form. Lead acid cell recharging- courtesy of By Sakar.solanki - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=23692756
|
Alkaline cell diagram courtesy of By Alkaline-battery.svg: *Schnitt_Alkali-Mn-Batterie.jpg: Tympanus.The original uploader was Tympanus at German Wikipediaderivative work: Tucvbif (talk)derivative work: Lead holder (talk) - Alkaline-battery.svg, Public Domain, https://commons.wikimedia.org/w/index.php?curid=15420424
CAR BATTERIES
•Lead-acid batteries are the most widely used secondary cells. •This is a car battery. •Although they appear to be a single unit, they are actually six separate cell connected together in a series. •Each cell contains three positive electrodes between four negative electrodes. •The electrodes are separated by a porous separator. •The positive electrodes consist of a lead grid packed with lead oxide. •Negative electrodes consist of a lead grid packed with powdered lead. •A solution of sulfuric acid acts as the electrolyte. •Each cell has a potential difference of just over 2 volts, a car battery has six of these so the total potential difference is about 12volts. •At the anode: Pb(s) + SO42-(aq) --> PbSO4(s) + 2e- •At the cathode: PbO2(s) + SO42-(aq) + 4H+(aq) + 2e- --> PbSO4(s)+ 2H2O(l) •The overall equation: Pb(s) + PbO2(s) + 2SO42-(aq) + 4H+(aq) ---> 2PbSO4(s) + 2H2O(l) The product of both electrode reactions forms a solid on the surface of the electrodes, enabling the battery to be recharged. •To recharge the battery, the electrode reactions are reversed. •The alternator is used to force electrons into the battery’s negative terminal and draw them out at the positive terminal. BATTERY LIFE •Battery life decreases over time due to changes within a battery. •Some factors affecting battery life are: -Loss of active materials (chemicals needed for reactions). -Impurities -Corrosion -Temperature (greater deterioration at higher temperatures). |
- Home
-
Unit 1
-
Unit 2
-
Unit 3
-
Unit 4
- Organic compounds >
- Reactions of organic compounds >
- Analysis of organic compounds-IR, NMR and MS >
- Analysis of organic compounds- Chromatography, HPLC >
- Analysis of organic compounds- volumetric analysis >
- Structure and bonding of food molecules >
- Metabolism of food in the human body >
- The energy content of food >
- Exam question links
- Scientific Reports and Posters
- Study score
- Chemistry basics
- Contact
- Resources
- Remote learning